By: Ronald A. Sherman, MD; Sharon Mendez, RN, CWS; and Catherine McMillan, BA
Maggot therapy is the controlled, therapeutic application of maggots to a wound. Simple to use, it provides rapid, precise, safe, and powerful debridement. Many wound care professionals don’t provide maggot therapy (also called wound myiasis) because they lack training. But having maggot therapy technology available for patients adds to your capabilities as a wound care provider.
Knowledge of maggot biology and life history helps wound care practitioners optimize therapy and anticipate or prevent problems. Educating patients and colleagues about maggot therapy can reduce stress and simplify your life as a wound care professional, whether you’re a novice or an experienced maggot therapist.
In this two-part series (the second part will appear in the November/December issue), the authors share their combined experience of more than 2,000 treatments to help you start and manage a maggot therapy service as part of a comprehensive wound care program.
Maggots as healers
The wound-healing benefits of maggots (fly larvae) have been documented for centuries. Wound care therapists have been placing maggots on wounds deliberately for at least 100 years—but flies have been depositing their larvae on wounds for at least 500 million years. That’s plenty of time for maggots to perfect their ability to dissolve necrotic tissue rapidly and stealthily without disturbing the host. (See From fly to maggot.)
Products
Myiasis refers to fly larvae living on a live vertebrate host. Larvae deposition in preexisting wounds isn’t always invasive. In fact, it may bring improvements in overall wound condition.
History and current status
One of the first clinicians to study and optimize maggot therapy was William Baer, MD, chief of orthopedic surgery at Johns Hopkins Hospital in Baltimore, Maryland. After the 1931 publication of Baer’s work, more than 1,000 surgeons incorporated maggot debridement therapy (MDT) into their practice; more than 90% reported being very satisfied with their experience. But by the mid-1940s, maggot therapy had nearly disappeared, at least in part due to the rise of antibiotics and the corresponding decrease in soft-tissue and bone infections.
By the late 1980s, the prevalence of antibiotic-resistant bacteria combined with the ability to keep patients with neurologic and cardiovascular impairments alive brought a resurgence in problematic soft-tissue wounds. The time was right to invite medicinal maggots back into hospitals and clinics.
Recent clinical trials irrefutably demonstrate the efficacy and safety of MDT. In fact, the pressing question for today’s wound care professionals isn’t “Does maggot therapy work?” but “How can I and my patients take advantage of this simple, effective, low-risk, low-cost technology?” (See Maggot mechanisms of action.)
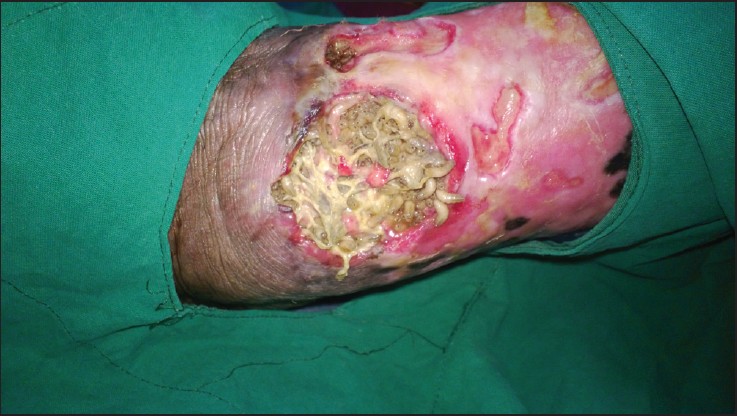
Whether using manufactured maggot-specific dressings or creating custom dressings at the bedside, clinicians have applied medicinal maggots to just about every external bodily surface imaginable except the eyes—from tiny wounds of nonhealing toe amputations to huge traumatic and burn injuries on the torso, extremities, and face; from fungating breast wounds to necrotic glans penis and resected Fournier’s gangrene of the perineal, genital, or perianal regions. Maggot dressings have even been applied to joints with infected hardware, the pleural cavity (via a persistently draining thoracotomy), and the necrotic peritoneum of an open abdomen. Almost any wound with nonviable soft tissue that’s open to the outside is a potential candidate for maggot therapy, although the larger and more complicated the topography, the harder it may be to confine the maggots. One caveat: Soft white, yellow, or tan slough is easy for newborn larvae to handle, whereas black dry eschar takes longer for the maggots to dissolve. Therefore, we always remove the eschar (through sharp debridement) or soften it before applying maggots. A simple autolytic dressing or, better yet, a wound gel under a hydrocolloid or thin film can be used to soften the eschar for a day or two while waiting for the maggots to arrive.
Therapeutic controls
Clinicians control or optimize myiasis so it’s most advantageous to the patient. Ways to control maggot therapy include:
- selecting species (usually Phaenicia [Lucilia] sericata) and strains proven to be safe and effective
- culturing contamination-free flies in laboratories
- disinfecting the maggots to remove microbes
- using quality-control assays to ensure the maggots are truly germ-free and larvae meet safety and regulatory standards.
Therapeutic controls continue at the bedside, as special dressings are used to maintain maggots on the wound during treatment.
Indications and contraindications
The Food and Drug Administration (FDA) has approved medicinal maggots for debridement of nonhealing necrotic skin and soft-tissue wounds. Commonly, such wounds include pressure ulcers, venous stasis ulcers, diabetic foot ulcers, and nonhealing traumatic or postsurgical wounds. Maggot therapy also has been used successfully in burns, in hospice care, and in debriding necrotic, fungating tumors. Sometimes, maggot therapy is used in conjunction with other modalities, such as systemic antibiotics, hyperbaric oxygen, and surgical debridement (for example, in osteomyelitis or necrotizing fasciitis).
Understanding maggot dressings
Because medicinal maggots feed only on necrotic tissue, they leave the wound when satiated or when all the necrotic tissue is gone. To prevent them from wandering away from the wound on their own, special dressings are used to confine them to the wound bed and its tributaries while letting air reach them (maggots are obligate oxygen breathers) and permitting efflux of wound exudate (maggots’ digestive enzymes liquefy the necrotic tissue). These dressings basically are maggot cages; the primary cage material usually is a fabric net.
The simplest dressings to apply are premade maggot cage dressings (for example, LeFlap™ and LeFlap DuJour™), which confine the maggots to simple planar wounds. Baglike dressings (such as LeSoc™) are useful for covering more complicated, nonplanar surfaces, such as toes, heels, and stumps. In Europe, maggots often are placed inside bags, which are sealed and laid over the wound (such as Biobag by BioMonde®). Although this containment dressing is easier to apply and remove (as long as the bag doesn’t break), studies show it leads to slower debridement, probably because the bag prevents larvae from directly accessing the entire wound bed, especially recesses and undermined crevices.
Making your own maggot dressing
While it’s easier to have a premade maggot dressing of the right size and shape, knowing how to make your own dressing at the bedside prepares you to use maggot therapy to any wound in any location. Attending a workshop or watching an experienced maggot therapist is a good way to learn how to make your own maggot dressing.
Below we describe our methods, which are widely used in North America. Above all, never forget your patient’s other needs—such as periwound skin protection, offloading, frequent turning, and good nutrition.
After evaluating and educating the patient and obtaining consent, we gently wash the wound (using only saline solution or water) and protect the periwound skin with a skin barrier or protectant. To keep maggots within the wound bed, we cover the periwound skin with a hydrocolloid pad or similar material. We cut the pad to surround, not cover, the wound; it then anchors the rest of the dressing. Thus, the pad functions as both a fence and a foundation on which to build the rest of the maggot cage.
For large wounds that a simple pad won’t cover, cut a hydrocolloid pad into strips and apply the strips to peripheral skin like a fence. Then place the maggots into the wound bed at a dose of 5 to 10 per cm2 of wound base. If you need only a few larvae, use a damp cotton swab to scoop them out. If larvae are available as maggot-impregnated gauze, simply remove the gauze from the jar and place the desired amount directly on the wound. If you need half the amount of maggots in the bottle, just use half of the maggot-impregnated gauze. If you’re not applying maggots within gauze, apply saline-moistened gauze loosely on top of the wound and maggots to provide scaffolding for them to crawl about.
Next, apply a layer of netting to serve as the top of the cage. You can use the foot of a panty hose, tight netting from a fabric store, or netted contact layer dressings (such as Wound Veil). Beware, though: Stretchable fabrics may let some of the tiny larvae escape when first applied, as larvae are less than 1 mm wide at that time. Instead, you can use a maggot-specific dressing made from fixed-weave polyester (for instance, Creature Comforts™ Polyester Net Dressing), which won’t stretch. Some therapists tape the fabric directly to the periwound skin or hydrocolloid barrier, but we find that gluing it to the hydrocolloid provides a more secure dressing.
Next, apply a latex-free glue, such as Nu-Hope Adhesive or LeGlue™, directly to the hydrocolloid before placing the net atop it, and apply another adhesive layer after the net is placed down, so the adhesive layers above and below the net meet and form a strong bond through the fabric pores. Topping the adhesive layer with fabric tape or zinc oxide tape or a transparent membrane dressing provides extra security. We extend this frame peripherally over the skin for dressings at risk of coming loose due to perspiration, soiling, or nearby flexion points, as well as for highly mobile patients. Constructing this custom cage dressing is more time-consuming than using a premade maggot dressing but allows you to apply medicinal maggots to any wound that needs them, almost anywhere on the body.
During maggot debridement, the larvae dissolve infected, necrotic tissue. Of course, this material will (and must) drain out of the porous cage. So we top the cage layer with light gauze or a similar absorbent material. The material should allow air into the cage dressing below and should be changed several times daily and whenever strikethrough occurs. If no drainage occurs, the wound (and larvae) may be too dry. In that case, add sterile saline solution, water, or irrigation fluid to the cage dressing. Some therapists routinely spray the dressing lightly with saline solution every 4 to 6 hours for the first 24 hours or top the cage dressing with a moistened gauze pad instead of dry gauze so the moisture wicks into the dressing below and hydrates the larvae.
With experience, optimizing and customizing maggot dressings becomes easier and faster. For a bedbound patient whose wound won’t be disturbed, you can simply ring the wound with zinc barrier paste and apply a breathable net on top to contain the maggots. Use ostomy paste to seal a crease in the skin where the hydrocolloid can’t seal.
Removing the dressing
Maggot dressings generally are removed after 48 hours. By that time, most maggots are satiated and ready to leave the wound. Sometimes the dressing is left on for 72 hours, but this increases the risk of a maggot breakout, because larvae that have finished feeding will try to pry the dressing off and escape. In some cases, the dressing is removed sooner than 48 hours—for example, if the patient reports pain that analgesics can’t adequately control.
Regardless of timing, the procedure for removing a maggot dressing is the same. Be prepared for fast-moving maggots trying to leave the wound as soon as you open the dressing. To make it easy to collect the larvae, place a biohazard bag around the wounded limb or tucked under the wounded buttock. Then loosen the tape and adhesives, peel back the cage dressing with one hand like a banana peel, and simultaneously wipe the maggots with a wet gauze pad in the other hand, sandwiching the maggots between your hands. In this way, most maggots won’t be seen and won’t escape. Those that manage to escape will drop into the plastic bag. If any maggots are still feeding or hiding, remove them by irrigation with water or saline solution or brush them off with a gauze pad or cotton swab. If some maggots are still holding on, simply cover them and the wound bed with a moist gauze pad. When the gauze pad is removed the next day, the last larvae will be done working and will be buried within the gauze pad.
Always discard maggot dressings in a plastic bag. Knot the top of the bag and throw it out with wound dressing waste. Double-bagging is best in case the outer bag rips from other items in the waste bin.
Applying and removing a maggot dressing sounds like a lot of work, but after you see the results of your first treatment, you’re likely to deem the effort worthwhile. By your fourth application, you should be able to apply a simple maggot dressing in just 5 or 10 minutes.
Managing pain and anxiety
The most common adverse events of maggot therapy are patient discomfort and patient (or provider) anxiety. Always read package inserts carefully before starting therapy. Take steps to prevent maggots and their secretions from contacting the intact periwound skin, which is susceptible to maceration, dermatitis, and cellulitis from prolonged exposure
to liquefied necrotic drainage. Also, the skin is highly sensitive to movement and pressure.
Maggots crawling over intact skin may feel like a caterpillar in the hand—tickly, itchy, or even unnerving. Most patients don’t feel pain within the wound bed, although some do complain of pain, especially with dressing changes. Such patients are likely to feel pain with maggot therapy, too. When larvae are about 24 hours old, they become large enough for the patient to feel as they crawl about the wound. Anticipate pain during maggot therapy in patients with underlying wound pain; use analgesics liberally to prevent or relieve it. Other ways to reduce pain are to administer fewer or smaller larvae (to increase the time before they can be felt) and to remove larvae early (because larvae grow larger and more active with time and are most uncomfortable when full-grown and trying to leave the wound bed).
Empowering patients with knowledge and control over their treatment helps decrease pain and anxiety. We routinely tell them that when analgesics no longer control pain to their satisfaction, we’ll remove the maggot dressings; all they have to do is ask. This puts them in control, reduces their anxiety, and increases their overall pain threshold. Of course, you have to make good on your promise. We ensure our patients have 24-hour access to someone who can assist immediately with dressing removal.
Occasionally, patients and therapists raise concerns about the foul odor arising as infected necrotic tissue liquefies. This is normal and generally occurs only at the start of treatment, when most of the decaying flesh is removed. Be aware that the smell comes from the patient’s fetid wound, not the maggots. Once most of the infected necrotic tissue has been removed, odor and drainage decrease dramatically or disappear altogether.
Coping with your own anxiety
Patients aren’t the only ones who may experience anxiety. Maggot therapy practitioners may become anxious, too. Anxiety about maggot therapy usually stems from ignorance, so become educated about maggot therapy. A great deal of educational material is available. You can read review articles on maggot therapy to learn about the basic principles and practices of maggot therapy. Visit the website of the BioTherapeutics, Education & Research (BTER) Foundation and attend conferences and workshops, such as the Wild on Wounds’ annual maggot therapy workshop. On its website, the BTER Foundation posts instructional videos on making custom-fit maggot dressings.
One of the most useful educational experiences is a one-on-one conversation with an experienced colleague or a former maggot therapy patient. In our practice, we encourage prospective patients to talk with previous patients. If this isn’t feasible, you can have prospective patients watch video interviews of past patients.
Many benefits, few risks
Maggot therapy is similar to other wound treatments in many respects, but different in a few ways. Medicinal maggots have been subjected to controlled trials of efficacy and safety, and their production and distribution are regulated by the FDA as single-use medical devices. But unlike most other devices, they are alive and therefore highly perishable. They can’t be stocked far ahead of use, and they have to be corralled during and after use.
Nonetheless, the similarities far outweigh the differences—just as the benefits of maggot therapy far outweigh the risks. When you add maggot therapy to your wound care toolbox, you’ll have more options for helping your patients. Access the many resources available to learn about maggot therapy and take advantage of every opportunity to practice your dressing skills. Finally, read this article again and again, if necessary, to help you advance quickly along your path to becoming not just a wise wound care therapist but also a skilled biotherapist.
Editor’s note: For information on setting up a maggot therapy service and providing patient education, read the September-October issue of Wound Care Advisor.
Selected references
Armstrong DG, Salas P, Short B, et al. Maggot therapy in “lower-extremity hospice” wound care: fewer amputations and more antibiotic-free days. J Am Podiatr Med Assoc. 2005;95(3):254-7.
Dumville JC, Worthy G, Bland JM, et al; VenUS II team. Larval therapy for leg ulcers (VenUS II): randomised controlled trial. BMJ. 2009;338:b773.
Fitzgerald RH, Armstrong DG. Palliative advanced wound care. Vein Magazine. 2009;2(2):44-6. www.veindirectory.org/magazine/article/palliative_advanced_wound_care. Accessed June 20, 2014.
Horobin AJ, Shakesheff KM, Pritchard DI. Promotion of human dermal fibroblast migration, matrix remodelling and modification of fibroblast morphology within a novel 3D model by Lucilia sericata larval secretions. J Invest Dermatol. 2006;126(6):1410-8.
Jukema GN, Menon AG, Bernards AT, Steenvoorde P, Rastegar AT, van Dissel JT. Amputation-sparing treatment by nature: “surgical” maggots revisited. Clin Infect Dis. 2002;35(12):1566-71.
Marineau ML, Herrington MT, Swenor KM, Eron LJ. Maggot debridement therapy in the treatment of complex diabetic wounds. Hawaii Med J. 2011; 70(6):121-4.
Markevich YO, McLeod-Roberts J, Mousley M, Melloy E. Maggot therapy for diabetic neuropathic foot wounds: a randomized study (abstr). European Association for the Study of Diabetes; 36th Annual Meeting; Jerusalem, Israel; 2000.
Mumcuoglu KY. Clinical applications for maggots in wound care. Am J Clin Dermatol. 2001;2(4):219-27.
Sherman RA. Maggot therapy for foot and leg wounds. Int J Low Extrem Wounds. 2002;1(2):135-42.
Sherman RA. Maggot therapy for treating diabetic foot ulcers unresponsive to conventional therapy. Diabetes Care. 2003;26(2):446-51.
Sherman RA. Maggot therapy takes us back to the future of wound care: new and improved maggot therapy for the 21st century. J Diabetes Sci Technol. 2009;3(2):336-44.
Sherman RA. Maggot versus conservative debridement therapy for the treatment of pressure ulcers. Wound Repair Regen. 2002;10(4):208-14.
Sherman RA. Mechanisms of maggot-induced wound healing: what do we know, and where do we go from here? Evid Based Complement Alternat Med. 2014;2014:592419. www.hindawi.com/journals/ecam/2014/592419/. Accessed June 20, 2014.
Sherman RA, Mumcuoglu KY, Grassberger M, Tantawi TI. Maggot therapy. In: Grassberger M, Sherman RA, Gileva OS, Kim CMH, Mumcuoglu KY, eds. Biotherapy—History, Principles and Practice: A Practical Guide to the Diagnosis and Treatment of Disease Using Living Organisms. Springer; 2013:5-29.
Sherman RA, Shapiro CE, Yang RM. Maggot therapy for problematic wounds: uncommon and off-label applications. Adv Skin Wound Care. 2007;20(11):
602-10.
Sherman RA, Wyle FA, Vulpe M. Maggot therapy for treating pressure ulcers in spinal cord injury patients. J Spinal Cord Med. 1995;18(2):71-4.
Steenvoorde P, Jacobi CE, Oskam J. Maggot debridement therapy: free-range or contained? An in-vivo study. Adv Skin Wound Care. 2005;18(8):430-5.
Steenvoorde P, Jacobi C, Wong C, Jukema G. Maggot debridement therapy in necrotizing fasciitis reduces the number of surgical debridements. Wounds. 2007;19(3):73-8.
Tantawi TI, Gohar YM, Kotb MM, Beshara FM, El-Naggar MM. Clinical and microbiological efficacy of MDT in the treatment of diabetic foot ulcers. J Wound Care. 2007;16(9):379-83.
Townley WA, Jain A, Healy C. Maggot debridement therapy to avoid prosthesis removal in an infected total knee arthroplasty. J Wound Care. 2006;15(2):78-9.
Wayman J, Nirojogi V, Walker A, Sowinski A, Walker MA. The cost effectiveness of larval therapy in venous ulcers. J Tissue Viability. 2000;10(3):91-4.
Wollina U, Liebold K, Schmidt WD, Hartmann M, Fassler D. Biosurgery supports granulation and debridement in chronic wounds—clinical data and remittance spectroscopy measurement. Int J Dermatol. 2002;41(10):635-9.
The authors work at the BioTherapeutics, Education & Research (BTER) Foundation in Irvine, California. Ronald A. Sherman is director. Sharon Mendez is a member of the board of directors. Catherine McMillan is a research intern.
Read part two here.
DISCLAIMER: All clinical recommendations are intended to assist with determining the appropriate wound therapy for the patient. Responsibility for final decisions and actions related to care of specific patients shall remain the obligation of the institution, its staff, and the patients’ attending physicians. Nothing in this information shall be deemed to constitute the providing of medical care or the diagnosis of any medical condition. Individuals should contact their healthcare providers for medical-related information.

Where and who does this in Ohio???????
Joyce, there is a referral page at the foundation where Dr. Sherman works: http://www.bterfoundation.org/maggotrx
It looks like you have to register (which appears to be free) to access it.
Cynthia Saver, MS, RN
Wound Care Advisor
My nam is Henry Meriweather I have had all of. My left toes amputated diou to diabetic wounds I ha another
Wound on my left fout bout I can’t find a wound treatment center that uses maggot therapy my email is
[email protected]