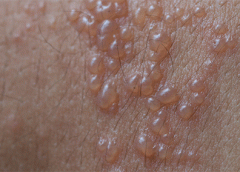
Herpes zoster (HZ, also called shingles) is a painful condition that produces a maculopapular and vesicular rash. Usually, the rash appears along a single dermatome (band) around one side of the body or face.
In most cases, pain, tingling, burning, or itching occurs a few days before the rash. Next, blisters form, scabbing over in 7 to 10 days. In rare cases, the rash is widespread, resembling varicella zoster (VZ, or chickenpox) rash. Pain can range from mild to severe and may be dull, burning, or gnawing. It may last weeks, months, or even years after the blisters heal. Shingles on the face may impair vision or hearing. (more…)
Read More