http://www.npuap.org/world-wide-pressure-ulcer-prevention-day-november-21-2013/
Read MoreSearch Results for: pressure
Clinical Notes: Pressure-Ulcer Data, Diabetic Foot Ulcers, IFG & HbA1c
Hospital pressure-ulcer comparison data not accurate
Performance scores for rates of hospital-acquired pressure ulcers might not be appropriate for comparing hospitals, according to a study in the Annals of Internal Medicine.
“Hospital report cards for hospital-acquired pressure ulcers: How good are the grades?,” funded by the Agency for Healthcare Research and Quality, analyzed 2 million all-payer administrative records from 448 California hospitals and quarterly hospital surveillance data from 213 hospitals from the Collaborative Alliance for Nursing Outcomes. (more…)
Read MoreDeveloping a cost-effective pressure-ulcer prevention program in an acute-care setting
By Tamera L. Brown, MS, RN, ACNS-BC, CWON, and Jessica Kitterman, BSN, RN, CWOCN
Pressure ulcers take a hefty toll in both human and economic terms. They can lengthen patient stays, cause pain and suffering, and increase care costs. The average estimated cost of treating a pressure ulcer is $50,000; this amount may include specialty beds, wound care supplies, nutritional support, and increased staff time to care for wounds. What’s more, national patient safety organizations and insurance payers have deemed pressure ulcers avoidable medical errors and no longer reimburse the cost of caring for pressure ulcers that develop during hospitalization. (more…)
Read MorePreventing pressure ulcers starts on admission
By Jeri Lundgren, BSN, RN, PHN, CWS, CWCN
The first 24 hours after a patient’s admission are critical in preventing pressure ulcer development or preventing an existing ulcer from worsening. A skin inspection, risk assessment, and temporary care plan should all be implemented during this time frame. Essentially, it’s the burden of the care setting to prove to insurers, regulators, and attorneys the pressure ulcer was present on admission and interventions were put into place to avoid worsening of the condition. Of course, patients also benefit from having their condition identified and treated promptly. (more…)
Read MoreApply QAPI to reduce pressure ulcer rates
By Jeri Lundgren, BSN, RN, PHN, CWS, CWCN
The Affordable Care Act of 2010 requires nursing homes to have an acceptable Quality Assurance and Performance Improvement (QAPI) plan within a year after the start of the QAPI regulation. While the implementation of this regulation may be a year out, now is the time to start applying its principles. Reducing pressure ulcer rates is a great program to target for a QAPI plan.
A team approach
If you decide to use pressure ulcers as your QAPI project, don’t take on your entire program at once. Break the program down into system subsets (for example, admission process, prevention program, and weekly rounds). Determining the status of your program in each subset—completed, needs improvement, or not completed—can help you prioritize which areas to target. It’s important you have support from leadership for your efforts.
I’ll use the example of the admission phase (ensuring that within the first 24 hours, skin and risk concerns are identified and a temporary plan of care is implemented) to illustrate a QAPI project. To address this area, a team was created, including representation from staff members involved with the admission process. The team then used the problem-solving model Plan-Do-Study-Act (PDSA) to examine the process.
The first step in the PDSA cycle is to Plan. During this step, you:
• evaluate and analyze the current process to determine baseline data, which are used to measure progress
• identify system performance gaps
• determine the root cause of the performance gaps
• develop an action plan that identifies the goals, steps, responsible staff, and target dates.
In our example, the team determined that within the first 24 hours, skin inspections were being completed only 10% of the time. The root-cause analysis revealed that the admission nurses didn’t feel competent to document identified pressure ulcers or skin concerns, so they deferred it until the wound nurse was available. The team’s action plan included the following:
• Develop and educate all the facility nurses on how to complete and accurately document a skin inspection.
• Develop and implement a competency evaluation to assess the nurse’s ability to apply the knowledge at the bedside.
• Develop an ongoing plan to ensure all nurses receive this education during orientation and yearly thereafter.
The team also set the following goal:
By the end of the next quarter, 100% of admitted patients will have an accurate skin inspection completed within 24 hours of admission.
The second step of the PDSA cycle is Do. During this step, you implement and execute the plan, while documenting your observations and recording data.
In our example, the “Do” was to:
• develop and provide the skin inspection education and bedside competency evaluations
• develop an evaluation and tracking
system
• add the education to the orientation program
• add the education to the staff development calendar to be offered yearly.
The third step of the PDSA cycle is to Study: In this phase, you:
• reevaluate and analyze the system
• compare the results with the baseline data and predictions
• summarize what was learned and accomplished and what needs to be improved
• determine if another PDSA cycle is
necessary to continue to improve the system.
Once all staff had been properly educated and competency testing completed, an analysis of the rate and accuracy of the admission skin inspections done within 24 hours of admission was completed. It was found that 100% of the patients admitted had a complete skin inspection done within 24 hours. However, not all the nurses could accurately stage pressure ulcers, so it was determined that the system needed improvement to ensure accurate assessments.
The last step of the PDSA cycle is to Act. In this step, you:
• determine what changes need to be made
• modify the plan to continue to improve the system
• repeat the PDSA cycle as necessary.
In our example, the team determined the nurses needed more guidance and education on staging of pressure ulcers. Therefore, a new PDSA cycle was set to ensure the nurses are competent in this area.
Benefits for staff and patients
It may be difficult to start the QAPI project and at times the process may be stressful, but keep in mind that a successful pressure ulcer QAPI project can improve not only the quality of life and care of your patients but also morale and team building for your staff. n
Jeri Lundgren is director of clinical services at Pathway Health in Minnesota. She has beenspecializing in wound prevention and management since 1990.
Read More
Assessing risk of pressure and moisture-related problems in long-term care patients
By Patricia A. Slachta, PhD, RN, ACNS-BC, CWOCN
Assessing moisture and pressure risk in elderly patients continues to be a focus for clinicians in all settings, particularly long-term care. Ongoing research challenges our ideas about and practices for cleansing and protecting damaged skin. Until recently, most wound care clinicians have cleansed long-term care patients’ skin with mild soap and water. But several studies have shown pH-balanced cleansers are more efficient than soap and water for cleansing the skin of incontinent patients.
Various terms are used to describe skin breakdown related to moisture—incontinence-associated dermatitis, perineal dermatitis, diaper rash, intertriginal dermatitis, intertrigo, moisture-related skin damage, moisture-associated skin damage, and even periwound dermatitis. This article uses moisture-associated skin damage (MASD) because it encompasses many causes of skin breakdown related to moisture. Regardless of what we call the condition, we must do everything possible to prevent this painful and costly problem.
Skin assessment
Start with an overall assessment of the patient’s skin. Consider the texture and note dryness, flaking, redness, lesions, macerated areas, excoriation, denudement, and other color changes. (See Identifying pressure and moisture characteristics by clicking the PDF icon above.)
Assessing MASD risk
A patient’s risk of MASD can be assessed in several ways. Two of the most widely used pressure-ulcer risk scales, the Norton and Braden scales, address moisture risk. The Norton and Braden subscales should drive your plan for preventing skin breakdown related to moisture or pressure. The cause of breakdown (moisture, pressure, or shear/friction) must be identified, because treatment varies with the cause.
Both the Norton and Braden scales capture activity, mobility, and moisture scores. The Braden scale addresses sensory perception, whereas the Norton scale identifies mental condition. (See Subscales identifying pressure, shear, and moisture risk by clicking the PDF icon above.) Also, be aware that two scales have been published for perineal risk, but neither has been used widely.
You must differentiate pressure- and moisture-related conditions to determine correct treatment. Patients who are repositioned by caregivers are at risk for friction or shear. Also, know that agencies report pressure-ulcer prevalence. Care providers no longer classify mucous-membrane pressure areas in skin prevalence surveys; mucous membranes aren’t skin and don’t have the same tissue layers. Furthermore, don’t report skin denudement from moisture (unless pressure is present) in prevalence surveys.
When moisture causes skin breakdown
Skin has two major layers—epidermis and dermis. The epidermis itself has five layers: The outermost is the stratum corneum; it contains flattened, keratin protein–containing cells, which aid water absorption. These cells contain water-soluble compounds called natural moisturizing factor (NMF), which are surrounded by a lipid layer to keep NMF within the cell. When skin is exposed to moisture, its temperature decreases, the barrier function weakens, and skin is more susceptible to pressure and friction/shear injury. Also, when urea in urine breaks down into ammonia, an alkaline pH results, which may reactivate proteolytic and lipolytic enzymes in the stool. (See Picturing moisture and pressure effects by clicking the PDF icon above.)
Caring for moisture-related skin breakdown
The standard of care for moisture-related skin breakdown includes four major components: cleanse, moisturize, protect, and contain. Specific products used for each component vary with the facility’s product formulary.
Cleanse
Gently wash the area using a no-rinse cleanser with a pH below 7.0. Don’t rub the skin. Pat dry.
Moisturize
Use creams containing emollients or humectants. Humectants attract water to skin cells and help hold water in the cells; don’t use these products if the skin is overhydrated. Emollients slow water loss from skin and replace intracellular lipids.
Protect
Options for skin protectants include:
• liquid film-forming acrylate sprays or wipes
• ointments with a petroleum, zinc oxide, or dimethicone base
• skin pastes. Don’t remove these products totally at each cleansing, but do remove stool, urine, or drainage from the surface and apply additional paste afterward. Every other day, remove the paste down to the bare skin using a no-rinse cleanser or mineral oil.
Be sure to separate skinfolds and use products that wick moisture rather than trap it. These may include:
• commercial moisture-wicking products
• a light dusting with powder containing refined cornstarch or zinc oxide—not cornstarch from the kitchen or powder with talc as the only active ingredient
• abdominal pads.
Contain
To keep moisture away from skin, use absorbent underpads with wicking properties, condom catheters (for males), fecal incontinence collectors, fecal tubes (which require a healthcare provider order), or adult briefs with wicking or gel properties. Call a certified ostomy or wound care nurse for tips on applying and increasing wear time for fecal incontinence collectors.
If 4″ × 4″ gauze pads or ABD pads are saturated more frequently than every 2 hours, consider applying an ostomy or specially designed wound pouch to the area. Collecting drainage allows measurement and protects skin from the constant wetness of a saturated pad.
Don’t neglect the basics, for example, know that wet skin is more susceptible to breakdown. Turn the patient and change his or her position on schedule. Change linens and underpads when damp, and consider using a low-air-loss mattress or bed or mattress with microclimate technology.
Also, be aware that fungal rashes should be treated with appropriate medications. If the patient’s skin isn’t too moist, consider creams that absorb into the skin; a skin-protecting agent can be used as a barrier over the cream. Besides reviewing and using the standards of care, you may refer to the Incontinence-Associated Dermatitis Intervention Tool, which has categories related to skin damage. See the “Incontinence-Associated Dermatitis Intervention Tool” (IADIT).
Bottom line on skin breakdown
To help prevent skin breakdown related to moisture, assess patients’ skin appropriately, determine treatment using evidence-based guidelines, and implement an appropriate plan of care.
Selected references
Black JM, Gray M, Bliss DZ, et al. MASD part 2: incontinence-associated dermatitis and intertriginous dermatitis: a consensus. J Wound Ostomy Continence Nurs. 2011;38(4):359-70.
Borchert K, Bliss DZ, Savik K, Radosevich DM. The incontinence-associated dermatitis and its severity instrument: development and validation. J Wound Ostomy Continence Nurs. 2010;37(5):527-35.
Doughty D. Differential assessment of trunk wounds: pressure ulceration versus incontinence-associated dermatitis versus intertriginous dermatitis. Ostomy Wound Manage. 2012;58(4):20-2.
Doughty D, Junkin J, Kurz P, et al. Incontinence-associated dermatitis: consensus statements, evidence-based guidelines for prevention and treatment, and current challenges. J Wound Ostomy Continence Nurs. 2012;39(3):303-15.
Gray M, Beeckman D, Bliss DZ, et al. Incontinence-associated dermatitis: a comprehensive review and update. J Wound Ostomy Continence Nurs. 2012;
39(1):61-74.
Gray M, Black JM, Baharestani MM, et al. Moisture-associated skin damage: overview and pathophysiology. J Wound Ostomy Continence Nurs. 2011;38(3):233-41.
Langemo D, Hanson D, Hunter S, Thompson P, Oh IE. Incontinence and incontinence-associated dermatitis. Adv Skin Wound Care. 2011;24(3):126-40.
National Pressure Ulcer Advisory Panel and European Pressure Ulcer Advisory Panel. Prevention and treatment of pressure ulcers: clinical practice guideline.Washington, DC: National Pressure Ulcer Advisory Panel; 2009.
Sibbald RG, Krasner DL, Woo KY. Pressure ulcer staging revisited: superficial skin changes & Deep Pressure Ulcer Framework©. Adv Skin Wound Care. 2011;24(12):571-80.
Wound, Ostomy and Continence Nurses Society. Guideline for Prevention and Management of Pressure Ulcers. Mt. Laurel, NJ: Wound, Ostomy and Continence Nurses Society; 2010.
Wound, Ostomy and Continence Nurses Society. Incontinence-Associated Dermatitis: Best Practice for Clinicians. Mt. Laurel, NJ: Wound, Ostomy and Continence Nurses Society; 2011.
Zulkowski K. Diagnosing and treating moisture-associated skin damage. Adv Skin Wound Care. 2012;25(5):231-6.
Patricia A. Slachta is an instructor at the Technical College of the Lowcountry in Beaufort, South Carolina.
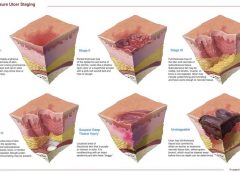
Read MoreFAQs for pressure ulcer staging
Why not call it a pressure ulcer?
By: Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
The most basic principle of healing a wound is to determine the cause—and then remove it. This is easier said than done, as many wounds have similar characteristics and we don’t always know all the facts leading up to the wound.
The process has been unnecessarily complicated by the recent pressure (no pun intended) to avoid at all costs calling a pressure ulcer a pressure ulcer. I use the term “unnecessarily” because it doesn’t matter what it’s called—a pressure ulcer, decubitus, “de-cube,” or bedsore—because in the end, the general idea is it’s bad news.
So what’s behind the desire to avoid calling it a pressure ulcer? First, a pressure ulcer has traditionally been equated to poor nursing care. As Florence Nightingale, the “Mother of Nursing,” wrote:
“If he has a bedsore, it’s generally not the fault of the disease, but of the nursing.”
No one likes to feel that he or she gave poor care, and as more hospital complications data are available to the public, reports of complications such as pressure ulcers affect people’s perceptions—right or wrong—about the care a hospital delivers.
The second reason gets at the “at all costs” part of the desire. The recent attention given to Medicare’s “present on admission” rule and “never” events has
elevated pressure ulcers high up the chain of “no-no’s” and put the hospital at risk for nonreimbursement. And many private insurers have followed Medicare’s lead in denying coverage for pressure ulcers that occur in the hospital. Unfortunately, all the focus on reimbursement is beginning to challenge even the best wound care experts, who simply want to get the patient’s wound healed.
Pressure from upper management has resulted in experts trying to bargain and rationalize their way out of calling it what it is (a pressure ulcer), instead calling it a bruise, not a deep-tissue injury. Or saying, “This is a shearing ulcer, not a pressure ulcer.” Or, my favorite: “It’s not an ischial pressure ulcer but a diabetic ulcer because the patient is a diabetic.” Wound care experts are being forced to question and doubt themselves because money, quality assurance, and reputation are on the line when an in-house wound is labeled a pressure ulcer.
Like crime scene investigation, determining wound etiology requires us to gather all the facts. Once the facts are in, systematically comparing and contrasting the clinical findings aids differential identification to pin down the type of wound present. It’s important that we assess and investigate all the following when searching for the cause:
• patient’s medical history
• recent activities (such as surgery, extensive X-rays, or long emergency-
department waits)
• comorbidities
• specific wound characteristics, such as location, distribution, shape, wound bed, and surrounding skin.
Naming the wound is an important first step in intervening. If the wound is caused by pressure, call it a pressure ulcer and jump into action. Remove the cause, heal the wound, and prevent further breakdown. Don’t let yourself be influenced by those who aren’t experts in wound care.
Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
Editor-in-Chief
Wound Care Advisor
Cofounder, Wound Care Education Institute
Plainfield, Illinois
Selected reference
Nightingale F. Notes on Nursing: What It Is, And What It Is Not. London: Harrison and Sons; 1859. http://ia600204.us.archive.org/17/items/notes
nursingnigh00nigh/notesnursingnigh00nigh.pdf.
Accessed August 30, 2012.
Get positive results with negative-pressure wound therapy
By Ronald Rock, MSN, RN, ACNS-BC
Complex wound failures are costly and time-consuming. They increase length of stay and contribute to morbidity and mortality in surgical patients. Negative-pressure wound therapy (NPWT)—a common adjunct to wound-care therapy—is used to accelerate wound healing in all fields of surgery. Using a vacuum device and wound-packing material, it applies subatmospheric pressure to complex wounds.
But NPWT alone doesn’t ensure adequate wound healing. Many physiologic factors—including infection, excessive moisture, nutrition, and medications—influence wound-healing success. Failure to account for these factors or improper application of NPWT can limit patient outcomes and cause debilitating complications.
For clinicians, applying and establishing an airtight seal on a complex wound is among the most dreaded, time-consuming, and challenging NPWT-related tasks. Simply applying NPWT material under layers of transparent drape may delay wound healing or exacerbate the wound. This article provides tips on safe application of NPWT to enhance the outcomes of patients with complex wounds.
Consider wound location
Wounds on the body’s anterior surfaces are less susceptible to the forces of pressure, friction, and shear than those on posterior and lateral surfaces. Posterior and lateral wounds commonly require posterior offloading or repositioning the patient in bed to reduce or eliminate direct pressure. This can be done with judicious and frequent patient turning using a specialty bed or support surface.
Bridge a posterior or lateral wound to an anterior surface by placing the drainage collection tubing to a nonpressure-bearing surface away from the wound. Bridging keeps the tubing from exerting pressure on intact skin and decreases the risk of a pressure ulcer. To create the bridge, cut foam into a single spiral of 0.5 to 1 cm, or if using gauze, fold gauze into 8 single layers.
Place the spiraled foam or gauze layers onto the drape, ensure the bridge is wider than the collection tubing disc, and secure it with an additional drape. Next, apply the NPWT collection tubing on the end of the bridge away from the wound. A wide bridge under the collection tubing disc will minimize the potential for periwound breakdown when negative pressure is initiated. You may modify this spiraling technique by varying the width of the foam to fill undermining and wounds of irregular configuration and depth.
Protect the periwound
An intact periwound may break down from exposure to moisture, injury from repetitive removal of a transparent drape, or NPWT material coming in contact with skin. Skin protection is critical in preventing additional breakdown stemming from contact with potentially damaging material.
Transparent drapes are designed to permit transmission of moisture vapor and oxygen. Avoid using multiple layers of transparent drapes to secure dressings over intact skin, as this can decrease the transmission of moisture vapor and oxygen, which in turn may increase the risk of fungal infection, maceration, and loss of an intact seal.
Periwound maceration also may indicate increased wound exudate, requiring an increase in negative pressure. Conversely, an ecchymotic periwound may indicate excessively high negative pressures. If either occurs, assess the need to adjust negative pressure and intervene accordingly. Reassess NPWT effectiveness with subsequent dressing changes.
Apply a protective liquid skin barrier to the periwound and adjacent healthy tissue to help protect the skin surface from body fluids. The skin barrier also helps prevent stripping of fragile skin by minimizing shear forces from repetitive or forceful removal of transparent drapes. Excessive moisture can be absorbed by using a light dusting of ostomy powder sealed with a skin barrier. A “window pane” of transparent drape or hydrocolloid dressing around the wound also can protect surface tissue from contactwith NPWT material and prevent maceration.
Avoid creating rolled wound edges
In the best-case scenario, epithelial tissue at the wound edge is attached to the wound bed and migrates across healthy granulation tissue, causing the wound to contract and finally close. With deep wound environments that lack moisture or healthy granulation tissue, the wound edges may roll downward and epibole may develop. Epibole is premature closure of the wound edges, which prevents epithelialization and wound closure when it comes in contact with a deeper wound bed. (See Picturing epibole by clicking the PDF icon above.)
Materials used in NPWT are primarily air-filled. Applying negative pressure causes air removal, leading to wound contraction by pulling on the wound edges—an action called macrostrain. Without sufficient NPWT material in the wound, macrostrain can cause the wound to contract downward and the wound edges to roll.
Ensure that enough NPWT material has been applied into the wound to enhance wound-edge approximation and avoid creating a potential defect as the wound heals. Before NPWT begins, material should be raised 1 to 2 cm above the intact skin. Additional material may be needed with subsequent changes if the NPWT material compresses below the periwound. The amount of NPWT material needed to remain above the periwound once NPWT starts varies with the amount of material compressed and the wound depth.
Reduce the infection risk
To some degree, all wounds are contaminated. Usually, the body’s immunologic response is able to clear bacterial organisms and wound healing isn’t delayed. But a patient who has an infection of a complex wound needs additional support.
Systemic antibiotics alone aren’t enough because they’re selective for specific organisms and don’t reach therapeutic levels in the wound bed. In contrast, topical antimicrobial adjuncts, such as controlled-release ionic silver, provide broad-spectrum antimicrobial coverage against fungi, viruses, yeasts, and gram-negative and gram-positive bacteria, including methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci.
Consider using controlled-release ionic silver for a wound known to be infected or at risk for infection due to its location or potential urine or fecal contamination. To be bactericidal, ionic silver must be in concentrations of at least 20 parts per million; also, it must be kept moist and must come in direct contact with infected wound bed. At lower concentrations, organisms may develop resistance. Ionic silver has no known resistance or contraindications. Dressings using it come in several forms, including a hydrogel sheet, perforated sheet, cavity version, and semiliquid hydrogel. Be sure the form you choose doesn’t occlude the NPWT material and compromise therapy. (See NPWT for a patient with necrotizing fasciitis by clicking the PDF icon above.)
View: NPWT
Obtain a negative-pressure environment
One of the most daunting aspects of NPWT is obtaining and maintaining a good seal—in other words, avoiding the dreaded leak. Preventive skin measures may contribute to a poor seal; skin-care products containing glycerin, surfactant, or dimethicone may prevent adequate adhesion of NPWT drapes. Body oil, sweat, and hair may need to be minimized or removed.
To avoid leaks, don’t overlook the obvious—loose connections, a loose drainage collection canister, exposed NPWT material, and skinfolds extending beyond the transparent drape. Tincture of benzoin (with or without a thin hydrocolloid dressing) increases tackiness to enhance the adhesive property of a transparent drape on the diaphoretic patient and on hard-to-drape areas, such as the perineum. But be sure to use tincture of benzoin with discretion, as it may remove fragile periwound tissue when the dressing is removed.
Ostomy paste products can serve as effective filler. These pliable products can be spread into position to obtain a secure seal under the transparent drape in hard-to-seal areas, such as the perineum. Pastes remain flexible and can be removed without residue. Temporarily increasing NPWT pressure to a higher setting may help locate a subtle leak or provide enough negative pressure to self-seal the leak. Once the leak resolves, remember to return the pressure to the ordered setting.
Knowledge optimizes healing
It’s important to be aware of potential complications of NPWT (See Take care with NPWT by clicking on the PDF icon above). However, when applied correctly, NPWT is an effective option for managing complex wounds. Recognizing and managing potential complications at the wound site, ensuring periwound protection, minimizing epibole formation, and preventing wound infection can result in a better-prepared wound bed and promote optimal healing.
View: NPWT case study
Selected references
Baranoski S, Ayello EA. (2012). Wound Care Essentials: Practice Principles. 3rd ed. Springhouse, PA; Lippincott Williams & Wilkins.
Bovill E, Banwell PE, Teot L, et al. Topical negative pressure wound therapy: a review of its role and guidelines for its use in the management of acute wounds. Int Wound J. 2008;5:511-529.
Sussman C, Bates-Jensen B. Wound Care: A Collaborative Practice Manual for Health Professionals. 4th ed. Baltimore, MD; Lippincott Williams & Wilkins; 2011.
Ronald Rock is an Adult Health Clinical Nurse Specialist in the Digestive Disease Institute at the Cleveland Clinic in Cleveland, Ohio.
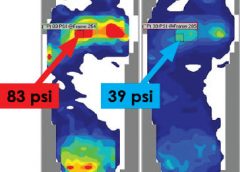
Read MorePressure mapping: A new path to pressure-ulcer prevention
By: Darlene Hanson, MS, RN, Pat Thompson, MS, RN, Diane Langemo, PhD, RN, FAAN, Susan Hunter, MS, RN, and Julie Anderson, PhD, RN, CCRC
Faced with the nursing diagnosis of Impaired skin integrity, we’ve all written care plans that state our goal as “redistributing or reducing pressure.” But how do we do that? Which measures do we take? And how do we know that our interventions have relieved pressure? Do we rely solely on a skin assessment? A patient’s self-assessment of comfort? What if the patient can’t feel pressure relief because of neurologic impairment?
The answers to these questions may be that nurses should use pressure mapping, a tool used by occupational and physical therapists to determine seat-interface pressures and by other healthcare professionals to perform foot assessments. (more…)
Read MoreRare ‘Flesh-Eating’ Bacterium Spreads North as Oceans Warm
Debbie King barely gave it a second thought when she scraped her right shin climbing onto her friend’s pontoon for a day of boating in the Gulf of Mexico on Aug. 13.
Even though her friend immediately dressed the slight cut, her shin was red and sore when King awoke the next day. It must be a sunburn, she thought.
But three days later, the red and blistered area had grown. Her doctor took one look and sent King, 72, to the emergency room.
Doctors at HCA Florida Citrus Hospital in Inverness, Florida, rushed King into surgery after recognizing the infection as Vibrio vulnificus, a potentially fatal bacterium that kills healthy tissue around a wound. While King lay on the operating table, the surgeon told her husband she would likely die if they didn’t amputate.
Just four days after the scrape, King lost her leg then spent four days in intensive care.
“The flesh was gone; it was just bone,” she said of her leg.
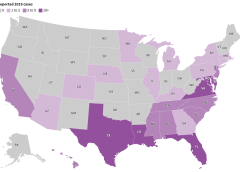
Cases of V. vulnificus are rare. Between 150 and 200 are reported to the Centers for Disease Control and Prevention every year, with about 20% resulting in death. Most are in states along the Gulf of Mexico, but, in 2019, 7% were on the Pacific Coast. Florida averages about 37 cases and 10 deaths a year.
But a rise in cases nationally and the spread of the disease to states farther north — into coastal communities in states such as Connecticut, New York, and North Carolina — have heightened concerns about the bacterium, which can result in amputations or extensive removal of tissue even in those who survive its infections. And warmer coastal waters caused by climate change, combined with a growing population of older adults, may result in infections doubling by 2060, a study in Scientific Reports warned earlier this year.
“Vibrio distributions are driven in large part by temperature,” said Tracy Mincer, an assistant professor at Florida Atlantic University. “The warmer waters are, the more favorable it is for them.”
The eastern United States has seen an eightfold increase in infections over a 30-year period through 2018 as the geographic range of infections shifted north by about 30 miles a year, according to the study, which was cited in a CDC health advisory last month.
The advisory was intended to make doctors more aware of the bacterium when treating infected wounds exposed to coastal waters. Infections can also arise from eating raw or undercooked seafood, particularly oysters, it warned. That can cause symptoms as common as diarrhea and as serious as bloodstream infections and severe blistered skin lesions.
New York and Connecticut this summer issued health warnings about the risk of infection as well. It’s not the first year either state has recorded cases.
“There’s very few cases but when they happen, they’re devastating,” said Paul A. Gulig, a professor in the Department of Molecular Genetics and Microbiology at the University of Florida College of Medicine.
‘An Accident of Nature’
Vibrio has more than 100 strains, including the bacterium that causes cholera, a disease that causes tens of thousands of deaths worldwide each year.
The V. vulnificus strain likes warm brackish waters close to shorelines where the salinity is not as high as in the open sea. Unlike some other Vibrio strains, it has no mechanism to spread between humans.
It’s found in oysters because the mollusks feed by filtering water, meaning the bacterium can become concentrated in oyster flesh. It can enter humans who swim in salty or brackish waters through the slightest cut in the skin. Infections are treated with antibiotics and, if needed, surgery.
“It’s almost an accident of nature,” Gulig said. “They have all these virulence factors that make them really destructive, but we’re not a part of this bug’s life cycle.”
Once inside the human body, the bacteria thrive.
Scientists don’t believe the bacteria eat flesh, despite how they’re often described. Rather, enzymes and toxins secreted by the bacterium as it multiplies break down the human tissue in the area below the skin, causing necrosis, or death of tissue cells.
The infection spreads like wildfire, Gulig said, making early detection critical.
“If you take a pen and mark where the edge of the redness is and then look at that two or four hours later, the redness would have moved,” Gulig said. “You can almost sit there and watch this spread.”
Researchers have conducted studies on the bacteria, but the small number of cases and deaths make it tough to secure funding, said Gulig. He said he switched his research focus to other areas because of the lack of money.
But growing interest in the bacteria has prompted talk about new research at his university’s Emerging Pathogens Institute.
Examining the bacteria’s genome sequence and comparing it with those of Vibrio strains that don’t attack human flesh could yield insights into potential drugs to interfere with that process, Gulig said.
Shock and Loss
Inside the operating room at HCA Florida Citrus, the only signs of King’s infection were on her shin. The surgeon opened that area and began cutting away a bright red mush of dead flesh.
Hoping to save as much of the leg as possible, the doctor first amputated below her knee.
But the bacteria had spread farther than doctors had hoped. A second amputation, this time 5 inches above the knee, had to be performed.
After surgery, King remained in critical care for four days with sepsis, a reaction to infection that can cause organs to fail.
Her son was there when she awakened. He was the one who told her she had lost her leg, but she was too woozy from medication to take it in.
It wasn’t until she was transferred to a rehab hospital in nearby Brooksville run by Encompass Health that the loss sank in.
A former radiation protection technician, King had always been self-reliant. The idea of needing a wheelchair, of being dependent on others — it felt like she had lost part of her identity.
One morning, she could just not stop crying. “It hit me like a ton of bricks,” she said.
Six different rehab staffers told her she needed to meet with the hospital’s consulting psychologist. She thought she didn’t need help, but she eventually gave in and met with Gerald Todoroff.
In four sessions with King, he said, he worked to redirect her perception of what happened. Amputation is not who you are but what you will learn to deal with, he told her. Your life can be as full as you wish.
“They were magic words that made me feel like a new person,” King said. “They went through me like music.”
Physical therapy moved her forward, too. She learned how to stand longer on her remaining leg, to use her wheelchair, and to maneuver in and out of a car.
Now, back in her Gulf Coast community of Homosassa, those skills have become routine. Her husband, Jim, a former oil company worker and carpenter, built an access ramp out of concrete and pressure-treated wood for their single-story home.
But she is determined to walk with the aid of a prosthetic leg. It’s the motivation for a one-hour regimen of physical therapy she does on her own every day in addition to twice-weekly sessions with a physical therapist.
Recovery still feels like a journey but one marked by progress. She has nicknamed her “stump” Peg. She’s now comfortable sharing before and after pictures of her leg.
And she’s made it her mission to talk about what happened so more people will learn about the danger.
“This is the most horrific thing that can happen to anybody,” she said. “But I’d sit back and think, ‘God put you here for a reason — you’ve got more things to do.’”
What to Know About ‘Flesh-Eating’ Bacterium Vibrio vulnificus
Infection Symptoms:
- Diarrhea, often accompanied by stomach cramping, nausea, vomiting, and fever.
- Wound infections cause redness, pain, swelling, warmth, discoloration, and discharge. They may spread to the rest of the body and cause fever.
- Bloodstream infections cause fever, chills, dangerously low blood pressure, and blistering skin lesions.
To Protect Against Vibrio Infections:
- Stay out of saltwater or brackish water if you have a wound or a recent surgery, piercing, or tattoo.
- Cover wounds with a waterproof bandage if they could come into contact with seawater or raw or undercooked seafood and its juices.
- Wash wounds and cuts thoroughly with soap and water after contact with saltwater, brackish water, raw seafood, or its juices.
Who Is Most at Risk:
- Anyone can get a wound infection. People with liver disease, cancer, or diabetes, and those over 40 or with weakened immune systems, are more likely to get an infection and have severe complications.
Sources:
- https://www.cdc.gov/vibrio/wounds.html
- https://my.clevelandclinic.org/health/diseases/24884-vibrio-vulnificus
This article was produced in partnership with the Tampa Bay Times.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
Read More
Bacterial biofilms, begone
By some estimates, bacterial strains resistant to antibiotics — so-called superbugs — will cause more deaths than cancer by 2050.
Colorado State University biomedical and chemistry researchers are using creative tactics to subvert these superbugs and their mechanisms of invasion. In particular, they’re devising new ways to keep harmful bacteria from forming sticky matrices called biofilms — and to do it without antibiotic drugs. (more…)
Read More