Category: Clinical Articles
Why not call it a pressure ulcer?
By: Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
The most basic principle of healing a wound is to determine the cause—and then remove it. This is easier said than done, as many wounds have similar characteristics and we don’t always know all the facts leading up to the wound.
The process has been unnecessarily complicated by the recent pressure (no pun intended) to avoid at all costs calling a pressure ulcer a pressure ulcer. I use the term “unnecessarily” because it doesn’t matter what it’s called—a pressure ulcer, decubitus, “de-cube,” or bedsore—because in the end, the general idea is it’s bad news.
So what’s behind the desire to avoid calling it a pressure ulcer? First, a pressure ulcer has traditionally been equated to poor nursing care. As Florence Nightingale, the “Mother of Nursing,” wrote:
“If he has a bedsore, it’s generally not the fault of the disease, but of the nursing.”
No one likes to feel that he or she gave poor care, and as more hospital complications data are available to the public, reports of complications such as pressure ulcers affect people’s perceptions—right or wrong—about the care a hospital delivers.
The second reason gets at the “at all costs” part of the desire. The recent attention given to Medicare’s “present on admission” rule and “never” events has
elevated pressure ulcers high up the chain of “no-no’s” and put the hospital at risk for nonreimbursement. And many private insurers have followed Medicare’s lead in denying coverage for pressure ulcers that occur in the hospital. Unfortunately, all the focus on reimbursement is beginning to challenge even the best wound care experts, who simply want to get the patient’s wound healed.
Pressure from upper management has resulted in experts trying to bargain and rationalize their way out of calling it what it is (a pressure ulcer), instead calling it a bruise, not a deep-tissue injury. Or saying, “This is a shearing ulcer, not a pressure ulcer.” Or, my favorite: “It’s not an ischial pressure ulcer but a diabetic ulcer because the patient is a diabetic.” Wound care experts are being forced to question and doubt themselves because money, quality assurance, and reputation are on the line when an in-house wound is labeled a pressure ulcer.
Like crime scene investigation, determining wound etiology requires us to gather all the facts. Once the facts are in, systematically comparing and contrasting the clinical findings aids differential identification to pin down the type of wound present. It’s important that we assess and investigate all the following when searching for the cause:
• patient’s medical history
• recent activities (such as surgery, extensive X-rays, or long emergency-
department waits)
• comorbidities
• specific wound characteristics, such as location, distribution, shape, wound bed, and surrounding skin.
Naming the wound is an important first step in intervening. If the wound is caused by pressure, call it a pressure ulcer and jump into action. Remove the cause, heal the wound, and prevent further breakdown. Don’t let yourself be influenced by those who aren’t experts in wound care.
Donna Sardina, RN, MHA, WCC, CWCMS, DWC, OMS
Editor-in-Chief
Wound Care Advisor
Cofounder, Wound Care Education Institute
Plainfield, Illinois
Selected reference
Nightingale F. Notes on Nursing: What It Is, And What It Is Not. London: Harrison and Sons; 1859. http://ia600204.us.archive.org/17/items/notes
nursingnigh00nigh/notesnursingnigh00nigh.pdf.
Accessed August 30, 2012.
Clinical Notes
New wound-swabbing technique detects more bacteria
The new Essen Rotary swabbing technique takes a few seconds longer to perform than traditional techniques, but improves bacterial count accuracy in patients with chronic leg ulcers, according to a study published by Wounds International.
“Evaluation of the Essen Rotary as a new technique for bacterial swabs: Results of a prospective controlled clinical investigation in 50 patients with chronic leg ulcers” reports that Essen Rotary detected significantly more bacteria compared to standard techniques and was the only one to identify five patients with methicillin-resistant Staphylococcus aureus (MRSA), compared to three detected by other techniques.
The Essen Rotary technique samples a larger surface area of the wound, which is beneficial for detecting MRSA.
“The Essen Rotary may become the new gold standard in routinely taken bacteriological swabs especially for MRSA screenings in patients with chronic leg ulcers,” the study authors write.
Reducing HbA1c by less than 1% cuts cardiovascular risk by 45% in patients with type 2 diabetes
A study presented at the American Diabetes Association 72nd Scientific Sessions found lowering HbA1c an average of 0.8% (from a mean of 7.8% to 7.0%, the treatment target) reduced the risk of cardiovascular death by 45% in patients with type
2 diabetes.
The absolute risk of mortality from a cardiovascular event was 9.9 events per 1,000 person-years in patients with decreasing HbA1c compared to 17.8 events in patients with stable or increasing HbA1c.
“HbA1c reduction and risk of cardiovascular diseases in type 2 diabetes: An observational study from the Swedish NDR” examined data from 18,035 patients in the Swedish National Diabetes Register.
CMS revises hospital, nursing home comparison websites
The Centers for Medicare & Medicaid Services (CMS) has enhanced two websites designed to help the public make informed choices about their health care.
Hospital Compare and Nursing Home Compare now have better navigation and new comparison tools. The two sites include data on quality measures, such as frequency of hospital-acquired infections, and allow the user to compare hospitals on these measures.
Improvements include easy-to-use maps for locating hospitals, a new search function that enables the user to input the name of a hospital, and glossaries that are easier to understand. It’s now also possible to access the data on the sites through mobile applications.
CMS maintains the websites, which are helpful for anyone who wants to compare facilities, not just patients on Medicare or Medicaid.
For more information, read the article in Healthcare IT News.
IOM releases report on accelerating new drug and diagnostics development
The Institute of Medicine (IOM) released “Accelerating the development of new drugs and diagnostics: Maximizing the impact of the Cures Acceleration Network—Workshop Summary.” The report is a summary of a forum that brought together members of federal government agencies, the private sector, academia, and advocacy groups to explore options and opportunities in the implementation of Cures Acceleration Network (CAN). The newly developed CAN has the potential to stimulate widespread changes in the National Institutes of Health and drug development in general.
Focus on individualized care—not just reducing swelling—in lymphedema patients
As a result of two extensive literature reviews, a researcher at the University of Missouri found that emphasizing quality of life—not just reducing swelling—is important for patients with lymphedema. Many providers and insurance companies base treatment on the degree of edema, but the volume of fluid doesn’t always correspond with the patients’ discomfort. Instead, an individualized plan of care should be developed.
The researchers found that Complete Decongestive Therapy (CDT), a comprehensive approach for treating lymphedema that includes skin and nail care, exercise, manual lymphatic drainage, and compression, may be the best form of specialized lymphedema management. For more information about CDT, watch for the November/December issue of Wound Care Advisor.
Plague case in Oregon draws national attention
An article about a case of the plague in Oregon has appeared on Huffington Post. A welder contracted the disease as a result of unsuccessfully removing a mouse from a stray cat’s mouth. Part of his hands have, in the words of the article, “darkened to the color of charcoal.” Later tests confirmed the cat had the plague.
Plague cases are rare in the United States. According to the Centers for Disease Control and Prevention, an average of 7 human cases are reported each year, with a range of 1 to 17 cases. Antibiotics have significantly reduced morality. About half of cases occur in people ages 12 to 45.
Use of negative pressure wound therapy with skin grafts
“Optimal use of negative pressure wound therapy for skin grafts,” published by International Wound Journal, reviews expert opinion and scientific evidence related to the use of negative pressure wound therapy with reticulated open-cell foam for securing split-thickness skin grafts.
The article covers wound preparation, treatment criteria and goals, economic value, and case studies. The authors conclude that the therapy has many benefits, but note that future studies are needed “to better measure the expanding treatment goals associated with graft care, including increased patient satisfaction, increased patience compliance and improved clinical outcomes.”
Mechanism for halting healing of venous ulcers identified
Researchers have identified that aberrantly expressed microRNAs inhibit healing of chronic venous ulcers, according to a study in The Journal of Biological Chemistry.
Six microRNAs were plentiful in 10 patients with chronic venous ulcers. The microRNAs target genes important in healing the ulcers. In an article about the study, one of the researchers said, “The more we know about the molecular mechanisms that contribute to [the development of venous ulcers], the more we can rationally develop both diagnostic tools and new therapies.”
Hemodialysis-related foot ulcers not limited to patients with diabetes
Both patients with diabetes and those without are at risk for hemodialysis-related foot ulcers, according to a study published by International Wound Journal.
Researchers assessed 57 patients for ulcer risk factors (peripheral neuropathy, peripheral arterial disease, and foot pathology, such as claw toes, hallux valgus, prominent metatarsal heads, corns, callosities, and nail pathologies) at baseline, and noted mortality 3 years later.
In all, 79% of patients had foot pathology at baseline, and 18% of patients without diabetes had peripheral neuropathy. Peripheral arterial disease was present in 45% of diabetic and 30% of nondiabetic patients. Nearly half (49%) of patients had two or more risk factors. Only 12% of patients had no risk factors. The presence of peripheral arterial disease and peripheral neuropathy increased risk of mortality.
The authors of “Prevalence of risk factors for foot ulceration in a general haemodialysis population” state that the high prevalence of risk factors in nondiabetic patients indicates that they are at risk for developing foot ulcers.
Study identifies risk factors for mortality from MRSA bacteremia
A study in Emerging Infectious Diseases found that older age, living in a nursing home, severe bacteremia, and organ impairment increase the risk of death from methicillin-resistant Staphylococcus aureus (MRSA) bacteremia.
Consultation with a specialist in infectious disease lowers the risk of death, and MRSA strain types weren’t associated with mortality.
“Predicting risk for death from MRSA bacteremia” studied 699 incidents of blood infection from 603 patients who had MRSA bacteremia.
Lymphedema 101 – Part 1: Understanding the pathology and diagnosis
By Steve Norton, CDT, CLT-LANA
Lymphedema is characterized by regional immune dysfunction, distorted limb contours, and such skin changes as papillomas, hyperkeratosis, and increased girth. The condition may involve the limbs, face, neck, trunk, and external genitals; its effects may include psychological distress. For optimal patient management, clinicians must understand what causes lymphedema and how it’s diagnosed and treated.
This two-part series provides an overview of lymphedema. Part 1 covers etiology, pathology, and diagnosis. Part 2, which will appear in the November-
December issue, will focus on treatment.
Causes of lymphedema
Lymphedema occurs when protein-rich fluid accumulates in the interstitium due to impaired lymphatic function. Proteins, other macromolecular wastes, and water constitute lymphatic loads. These wastes rely on specially structured absorptive and transport structures in peripheral regions for their return to central circulation.
When lymph stasis prevails, inflammatory processes and lymphostatic fibrosis trigger tissue-density changes, further entrapping superficial vessels and accelerating mechanical insufficiency. (See Physiologic changes caused by lymphatic disruption by clicking the PDF icon above.)
Classifying lymphedema
Lymphedema can be primary or secondary. Primary lymphedema either is congenital (present at birth) or arises around puberty. In the vast majority of cases, it is associated with structural changes in the lymphatic system and isn’t associated with another disease or condition. Most structural changes (87%) manifest before age 35 and cause hypoplasia of vessels and nodes. Syndromes involving hyperplasia, node fibrosis, or aplasia also may occur, although they’re much less common. Dysplasia (either hypoplasia, hyperplasia, or aplasia) predisposes drainage regions to inadequate lymph collection, resulting in edema and secondary tissue changes, such as chronic inflammation and reactive fibrosis. Genetic variability in lymphatic constitution may explain why seemingly similar patients receiving the same surgical protocol have different lymphedema risks over time.
Secondary lymphedema stems from a significant insult to lymphatic tissues, as from lymphadenectomy, radiation therapy, trauma, infection, or cancer. It commonly results from direct trauma to regional nodes or vessel structures. Slow degradation of lymphatic function also occurs when adjacent tissues (such as superficial and deep veins) become diseased, when cellulitis occurs, or when accumulations
of adipose or radiation fibrosis mechanical-ly disrupt drainage of skin lymphatics.
Lymphedema stages
Lymphedema progresses in stages, which involve secondary connective-tissue disease combined with disturbed fluid update and transport. These conditions cause a universal and classic clinical picture.
• Stage 0 (latency stage) is marked by reduced transport capacity and functional reserve. The patient has no visible or palpable edema, but has such subjective complaints as heaviness, tightness, and waterlogged sensations.
• In Stage 1 edema (reversible lymphedema), edema decreases with elevation. Pitting edema is present, but fibrosis is absent.
• During Stage 2 (spontaneously irreversible lymphedema), lymphedema doesn’t resolve entirely, although it may fluctuate. Pitting is more pronounced and fibrosis is present.
• Stage 3 (lymphostatic elephantiasis) is marked by dermal hardening, nonpitting edema, papillomas, hyperkeratosis, and in some cases, extreme girth.
Assessment and diagnosis
Diagnosing lymphedema can be challenging because edema may be associated with other diseases and disorders. For a summary of signs and symptoms, see Clinical findings in lymphedema by clicking the PDF icon above.
Discomfort and skin appearance
Lymphedema rarely causes pain because the skin accommodates gradual, insidious fluid accumulation. However, secondary orthopedic discomfort may result from increased weight of the affected limb due to deconditioning or decreased range of motion.
Because lymphedema usually progresses slowly, gravity and centrifugal forces pull fluids toward distal limb areas, causing an entrenched, stubborn pitting edema. Later, further valvular incompetence contributes to worsening distal edema in the fingers, toes, and dorsal regions of the hand and foot. Prominent lower-extremity structures, such as the malleolus, patella, tibia, anterior tibialis tendon, and Achilles tendon, become progressively less distinct. This creates a columnar limb appearance; the swollen limb has the same girth from distal to proximal aspects, unlike the natural cone shape of a normal limb.
Lymphatic failure doesn’t tax the venous system, so skin color remains normal. Blood supply remains patent, helping to prevent secondary ulcers.
Severity
Lymphedema severity correlates directly with such factors as onset of the condition and extent of cancer therapy, if given (number of nodes resected, number of positive nodes, and use of radiotherapy). Lymphedema may worsen with a greater number of infection episodes, weight gain, injury, diuretics, limb disuse, pneumatic compression therapy (when used for pure lymphedema), and ill-fitting compression garments. The single most important contributor to increasing lymphedema severity is lack of patient education, which can result in improper treatment or none at all.
Opportunistic infections
Lymphedema causes regional immune suppression and leads to an increase in opportunistic infections such as cellulitis. As skin integrity suffers, scaling and dryness allow resident skin pathogens (such as streptococci and staphylococci) to gain access through the defective skin barrier into protein-rich interstitial fluid, creating a medium favorable to bacterial colonization. Lymphocyte migration decreases, and dissected or irradiated nodal sites are slow to detect invaders. Furthermore, stagnant lymph promotes further delays in the immune response. Patients with opportunistic infections may exhibit high fever, local erythema, regional hypersensitivity or acute pain, flulike symptoms, and rapidly advancing “map-like” borders in the skin.
Differential diagnosis
Several methods can aid differential diagnosis.
Clinical findings. Lymphedema can be diagnosed from patient history, physical examination, palpation, and inspection. Trauma to lymph nodes (each of which governs a distinct body region) decreases the transport capacity of lymph formed in that region, in turn causing local swelling (lymphedema). Trauma to the axillary or inguinal lymph nodes, which exist on both the left and right of the body and in both the upper and lower regions, predisposes these quadrants to swelling. Therefore, if lymph nodes on only one side are damaged, lymphedema occurs only on that side of the body. Using the universal characteristics cited above as a guide, while ruling out cancer recurrence, acute deep vein thrombosis, or plasma protein abnormalities, yields sufficient data to form a diagnosis.
Imaging. Lymphography involves subcutaneous injection of a lymph vessel–
specific dye (Patent Blue V), followed by X-ray. Although it provides high-resolution images of lymphatic structures, this technique is invasive, painful, damaging to lymphatics, and potentially lethal—and therefore is no longer recommended.
Lymphangioscintigraphy (LAS) uses interdigital subcutaneous injection of protein-labeled radioisotopes, followed by
imaging at specific intervals to gather information about uptake and transport time. Images are hazy and false-negatives are common, so well-trained radiotherapists familiar with lymphology and lymphedema should administer and interpret the test. Also, experts don’t agree on standard criteria for LAS administration, so measures may not be similarly conclusive.
Limb-measuring instruments and methods. Serial measurement of affected limb circumference using a standard garment tape measure is the most widely accessible approach. Intra-rater reliability is comparable to that of currently used tools; however, these methods can’t be used for early detection, for screening, or when various raters are used to assess the same patient. Circumferences are measured at four points and are considered positive if a distance of 2 cm or more separates the involved from uninvolved extremity in comparison. Water displacement techniques for limb-volume calculation, although accurate, are impractical in most clinical settings and rarely used.
Various devices have been used to obtain measurements. For instance, the Perometer® uses optoelectronic volumetry. By scanning the limb with infrared beams circumferentially, the device accurately records girth at 4-mm intervals along the limb length and transmits these measurements to a computer. The Perometer is used mainly in the research setting. Preoperative and postoperative measurements at intervals can detect lymphedema early.
Impedimed XCA® uses bioelectrical
impedance to calculate ratios of intracellular to extracellular fluid. A weak electrical current is passed through affected and unaffected limbs, allowing comparison of results. Impedance is lower in edematous tissue, supporting an accurate diagnosis.
Next step: Treatment
Once a diagnosis is made, the next step is treatment. Part 2 of this series covers lymphedema treatment.
Selected references
Foeldi M. Foeldi’s Textbook of Lymphology: For Physicians and Lymphedema Therapists. 3rd ed. St. Louis, MO: Mosby; 2012.
Kubik S, Manestar M. Anatomy of the lymph capillaries and precollectors of the skin. In: Bollinger A, Partsch H, Wolfe JHN, eds. The Initial Lymphatics. Stuttgart: Thieme-Verlag; 1985:66-74.
Lee B, Andrade M, Bergan J, et al. Diagnosis and treatment of primary lymphedema. Consensus document of the International Union of Phlebology (IUP)—2009. Int Angiol. 2010 Oct;29(5):454-70.
Lerner R. Chronic lymphedema. In: Prasad H, Olsen ER, Sumpio BE, Chang JB, eds. Textbook of Angiology. Springer; 2000.
Mayrovitz HN. Assessing lymphedema by tissue indentation force and local tissue water. Lymphology. 2009 June;42(2):88-98
National Cancer Institute. Lymphedema (PDQ®): Health Professional Version. Updated June 30, 2011. www.cancer.gov/cancertopics/pdq/supportivecare/
lymphedema/healthprofessional. Accessed September 5, 2012.
Northrup KA, Witte MH, Witte CL. Syndromic classification of hereditary lymphedema. Lymphology. 2003 Dec:36(4):162-89.
Olszewski WL. Lymph Stasis: Pathophysiology, Diagnosis and Treatment. CRC Press; 1991.
Pecking AP, Alberini JL, Wartski M, et al. Relationship between lymphoscintigraphy and clinical findings in lower limb lymphedema (LO): toward a comprehensive staging. Lymphology. 2008 Mar;41(1):1-10.
Stanton AW, Northfield JW, Holroyd, B, et al. Validation of an optoelectronic volumeter (Perometer). Lymphology. 1997 June;30(2):77-97
Weissleder H, Schuchhardt C. Lymphedema: Diagnosis and Therapy. 4th ed. Viavital Verlag GmbH; 2007.
Steve Norton is cofounder of Lymphedema & Wound Care Education and executive director of the Norton School of Lymphatic Therapy in Matawan, New Jersey.
Read MoreTeaching ostomy patients to regain their independence
By Goranka Paula Bak, BSN, RN, ET, CWOCN
Every year, thousands of people of all ages have ostomy surgery in the United States. That means no matter where you work, you’re likely to care for patients with new ostomies and teach them to care for themselves. (See Why patients need ostomy surgery by clicking the PDF icon above.)
These patients present two challenges: staying up-to-date on ostomy equipment and procedures and having only 3 to 5 days after surgery to teach basic ostomy care to patients who are unlikely to be physically or emotionally ready to learn. This review and update of ostomy care should help you meet both challenges. Let’s start with the common types of stomas and ostomies.
Reviewing stoma types
The three common types of stomas are the end stoma, loop stoma, and double-barrel stoma. The patient’s surgeon decides on the most appropriate type based on the disease or injury, the amount of intestine available, and the patient’s health status.
To create an end stoma, the most common type, a surgeon creates a small opening in the abdominal wall, then resects the intestine and pulls it through the opening. Next, the surgeon turns the end of the intestine onto itself—somewhat like cuffing a sock—and sutures the stoma to the skin. Next to the stoma, the surgeon may create a mucous fistula, which secretes mucus from the nonfunctioning portion of the intestine.
A loop stoma is usually created for a temporary ostomy. The surgeon creates a small opening in the abdominal wall and pulls a loop of intestine through it. With an external rod or bridge placed under the loop to keep it from slipping back into the peritoneal cavity, the surgeon cuts the top of the loop to form the apex of the stoma. Next, the surgeon everts the intestine around the opening and sutures the stoma edges to the skin. After a few days, the rod is removed, and the stoma stays above skin level on its own.
Double-barrel stomas aren’t common in adults, but they are used frequently in neonates. For this type, a surgeon cuts through the bowel to create two separate end stomas, one to evacuate stool and the other to evacuate mucus. The two stomas can be placed next to each other, so one pouching system can be used for both. If the stomas are further apart, the patient may need two pouches. If mucus secretions are slight, you may be able to apply a nonadherent dressing after gently cleaning the stoma and peristomal skin, instead of using a pouch.
An ideal stoma protrudes above the skin surface and has a flat peristomal skin surface that allows the ostomy barrier to adhere to it. (See Characteristics of an ideal stoma by clicking the PDF icon above.) A healthy stoma is red and moist and looks like gum tissue. In fact, the entire alimentary canal from mouth to anus has the same type of tissue. Keep in mind that while cleaning a stoma, you’ll commonly see a small amount of bleeding, just as your gums may bleed when you brush your teeth too vigorously. If you note excessive bleeding, report it immediately.
Reviewing types of ostomies
The three most common types of ostomies you’ll see are a colostomy, an ileostomy, and a urostomy.
A surgeon creates a colostomy by making an opening in the large intestine and connecting it to the stoma. The location of the surgical opening will affect the consistency of the stool exiting the body through the stoma: The lower the opening, the firmer the consistency. A colostomy produces flatus because of the high bacterial content in the large intestine. A colostomy can be temporary or permanent, depending on the diagnosis and the patient’s ability to heal.
To create an ileostomy, a surgeon makes an opening in the small intestine and forms a stoma. Patients with ileostomies are prone to dehydration because a large amount of enzymatic fluid is discharged through the ileostomy instead of being absorbed in the disconnected large intestine. Teach your patient to be alert for the signs and symptoms
of dehydration—thirst; decreased urination; darker, concentrated urine; warm, flushed skin; fever; and malaise. Also, warn the patient to keep enzymatic effluent off the skin because it can quickly cause skin breakdown. As with a colostomy, an ileostomy may be temporary or permanent.
To create a urostomy, a surgeon removes the diseased bladder and uses a portion of resected small intestine to create a conduit. The ureters, which were connected to the bladder, are sutured into the resected small intestine so urine can exit the body through the stoma. Urostomies are permanent.
Choosing the right pouching system
An ostomy pouching system has two parts: an adhesive barrier that attaches to the skin around the stoma and a pouch connected to the barrier that collects effluent. Determining the right ostomy pouching system for your patients will depend on their needs and preferences. Let’s look at some of the important choices.
A pouching system may come in one or two pieces. With one-piece systems, the barrier (also called the wafer, flange, or baseplate) and pouch come as a single unit. (See One- and two-piece ostomy pouching systems by clicking the PDF icon above.) One-piece systems are easier to apply and more flexible. Plus, they’re flatter and thus more discreet.
With two-piece systems, the barrier and pouch are separate components that must be attached. Depending on the product, the patient may need to press the pouch onto the barrier or use a locking ring mechanism or an adhesive coupling system. Two-piece systems allow the patient to remove the pouch without removing the barrier. Also, a patient can apply a smaller pouch, such as a stoma cap or close-ended pouch, before exercise or intimate relations.
Today, ostomy pouching systems offer many options that weren’t available just a few years ago—options such as built-in, varying levels of convexity and integrated closures. Skin-friendly barriers now eliminate the need for accessory products, such as skin prep, ostomy powder, and stoma paste. By knowing the new options, you can help simplify the pouch application process for your patient and increase patient satisfaction.
Selecting barriers
Barriers may be cut-to-fit or precut. Cut-to-fit barriers are used during the postoperative period while the stoma size is changing and for oval or irregularly shaped stomas. Precut sizes are convenient for round stomas after they stabilize.
The choice of a barrier also depends on abdominal contours and the effluent consistency. A standard-wear barrier is appropriate for thicker output. An extended-wear barrier is better for loose or watery output and for problematic ostomies.
Convex barriers can be used to push down the peristomal skin and help the stoma project into the pouch. These barriers help ensure that the effluent goes into the pouch and not between the skin and the barrier, causing leakage.
For ostomies that are difficult to manage and frequently leak, you may select an ostomy belt. This device helps secure a convex pouching system by increasing its pressure. The plastic hooks at the end of the belt attach to the belt loops of the pouching system. Depending on the manufacturer, the belt loops may be on the barrier or the pouch.
Selecting pouches
Pouches can be transparent or opaque. Transparent pouches allow you to see the stoma postoperatively and allow patients to watch as they place the pouch over the stoma. Opaque pouches, of course, have the advantage of concealing the effluent.
Some pouches have filters. Colostomy patients usually prefer filtered pouches because they eliminate the need to burp the pouch to remove gas. Ileostomy output is usually watery, so the charcoal filters may get wet and quit working.
The appropriate type of pouch closure also varies, depending on the type of output and the patient’s needs and preferences. A patient with a colostomy or ileostomy needs a drainable pouch. Newer clamps and integrated closures can make closing the pouch easier for the patient.
Close-ended pouches are available for patients who empty their pouch once or twice a day. Reimbursement guidelines for Medicare, which most insurance companies follow, allow 60 close-ended pouches a month. Thus, someone who empties once or twice a day will have enough pouches.
Patients with urostomies use pouches with spigots on the end to allow for urine drainage.
Urostomy pouches can be connected to a continuous urinary drainage bag at bedtime or to a leg bag during the day for those in a wheelchair.
Dealing with complications
Report the following postoperative complications to the surgeon and the ostomy clinician caring for the patient:
• allergic reaction
• candidiasis
• contact dermatitis
• folliculitis
• ischemic or necrotic stoma
• mechanical irritation
• mucocutaneous separation
• parastomal hernia
• prolapse.
Many complications result from surgical technique or the patient’s disease status, but complications also result from an incorrect fit or an ostomy that frequently leaks. To determine why and where leakage occurred, examine the back of the barrier. If leakage results from a flush or recessed stoma or an irregular surface around the stoma, the patient may need a convex barrier. Or the patient may need a convex barrier and an ostomy belt.
Remember that a firm abdomen requires a soft, flexible barrier that conforms to the abdominal contours. A soft belly, on the other hand, may need the rigidity of a convex barrier to help the stoma empty into the pouch.
For weepy, extremely denuded skin, apply ostomy powder, brush off the excess, and dab an alcohol-free skin sealant, such as No-Sting by 3M, on the area. Allow the area to air-dry before applying the barrier.
Discharge planning and patient teaching
Because the postoperative length of stay is short, start preparing for discharge and teaching the pouch-change procedure at your first hospital encounter with the patient. Teaching the patient at each encounter creates a continuum of care throughout hospitalization and recovery. If possible, include the caregiver. (See Teaching your patient to change an ostomy pouch by clicking the PDF icon above.) Before discharge, give the patient two or three ostomy pouches to take home, step-by-step printed instructions on applying pouches, a list of supplies the patient will need, and a list of ostomy product suppliers in the area.
Also, teach the patient about complications, activity, diet, and drug therapy and provide the names of local support groups. (See Finding support by clicking the PDF icon above.)
Complications at home
Tell the patient to call the physician if any of the following occurs: fever, increased pain or discomfort, diarrhea, dehydration, or signs and symptoms of infection at the surgical site. The patient should also call the physician if there’s no output from the stoma for more than 24 hours.
Patients with urostomies who develop the following signs and symptoms may have a urinary tract infection: fever, chills, abdominal or retroperitoneal pain, and bloody, cloudy, or foul-smelling urine. Clear urine with mucus shreds is normal.
Increasing activity
Explain that after surgery the patient should feel a little bit stronger every day. Teach the patient to alternate rest and activity periods and to increase the activity period a little each day while building endurance. Refer the patient to the physician for clearance to resume exercise and sexual relations.
Dietary considerations
After ostomy surgery, your patient will start on a clear liquid diet and progress to a normal, preoperative diet. The patient should reintroduce one food at a time to help determine if it’s well tolerated. High-fiber foods should be introduced slowly.
Tell your patient to expect his or her body to tolerate the food as it did before surgery: if onions caused gas before surgery, they will do so after surgery, too, but the stoma will expel the gas. Make the patient aware of other gas-forming foods, such as hard-boiled eggs, spicy foods, fish, dried beans, carbonated beverages, and beer.
Tell the patient to chew food well and to drink plenty of liquids, which will prevent blockage. Explain the importance of hydration to avoid constipation, dehydration, and urinary tract infection. To prevent dehydration, ileostomy patients who are not on fluid restriction should drink 64 ounces of fluid a day. Teach these patients to replace fluids by drinking a glass of water every time they empty their pouches.
Drug therapy
Tell the patient to adhere to the prescribed regimen and to call the physician before using any new drugs. Instruct patients with ileostomies not to take laxatives because they may become dehydrated. These patients should tell their pharmacist that they have an ileostomy, so the proper form of prescription drugs can be ordered. Extended-release tablets will not break down in time and may be found in the pouch.
Rewarding work
Keeping up with the changes in ostomy equipment and care and teaching new ostomy patients the skills they need can be challenging. But helping these patients regain the confidence and learn the skills they need to improve their quality of life makes the rewards much greater than the challenges.
Selected references
Colwell J. Principles in stoma management. In: Colwell JC, Goldberg MT, Carmel JE, eds. Fecal and Urinary Diversions Management Principles. St. Louis, MO: Mosby; 2004:240-262.
Discharge Planning for a Patient with a New Ostomy: Best Practice for Clinicians. Glenview, IL: Wound, Ostomy and Continence Nurses Society; 2004.
Hampton BG, Bryant RA. Ostomies and Continent Diversions: Nursing Management. St. Louis, MO: Mosby; 1992.
McCann E. Common ostomy problems. In: Milne C, Corbett L, Dubuc D. Wound, Ostomy, and Continence Nursing Secrets: Questions and Answers Reveal the Secrets to Successful WOC Care. Philadelphia, PA: Hanley & Belfus, Inc; 2004.
The Sarah Cole Hirsh Institute for Best Nursing Practices Based on Evidence. State of the evidence review hospital to home: Planning the transitions. 2000; Cleveland, OH.
Goranka Paula Bak is an Ostomy Sales Territory Manager for Coloplast Corporation.
Read MoreGet positive results with negative-pressure wound therapy
By Ronald Rock, MSN, RN, ACNS-BC
Complex wound failures are costly and time-consuming. They increase length of stay and contribute to morbidity and mortality in surgical patients. Negative-pressure wound therapy (NPWT)—a common adjunct to wound-care therapy—is used to accelerate wound healing in all fields of surgery. Using a vacuum device and wound-packing material, it applies subatmospheric pressure to complex wounds.
But NPWT alone doesn’t ensure adequate wound healing. Many physiologic factors—including infection, excessive moisture, nutrition, and medications—influence wound-healing success. Failure to account for these factors or improper application of NPWT can limit patient outcomes and cause debilitating complications.
For clinicians, applying and establishing an airtight seal on a complex wound is among the most dreaded, time-consuming, and challenging NPWT-related tasks. Simply applying NPWT material under layers of transparent drape may delay wound healing or exacerbate the wound. This article provides tips on safe application of NPWT to enhance the outcomes of patients with complex wounds.
Consider wound location
Wounds on the body’s anterior surfaces are less susceptible to the forces of pressure, friction, and shear than those on posterior and lateral surfaces. Posterior and lateral wounds commonly require posterior offloading or repositioning the patient in bed to reduce or eliminate direct pressure. This can be done with judicious and frequent patient turning using a specialty bed or support surface.
Bridge a posterior or lateral wound to an anterior surface by placing the drainage collection tubing to a nonpressure-bearing surface away from the wound. Bridging keeps the tubing from exerting pressure on intact skin and decreases the risk of a pressure ulcer. To create the bridge, cut foam into a single spiral of 0.5 to 1 cm, or if using gauze, fold gauze into 8 single layers.
Place the spiraled foam or gauze layers onto the drape, ensure the bridge is wider than the collection tubing disc, and secure it with an additional drape. Next, apply the NPWT collection tubing on the end of the bridge away from the wound. A wide bridge under the collection tubing disc will minimize the potential for periwound breakdown when negative pressure is initiated. You may modify this spiraling technique by varying the width of the foam to fill undermining and wounds of irregular configuration and depth.
Protect the periwound
An intact periwound may break down from exposure to moisture, injury from repetitive removal of a transparent drape, or NPWT material coming in contact with skin. Skin protection is critical in preventing additional breakdown stemming from contact with potentially damaging material.
Transparent drapes are designed to permit transmission of moisture vapor and oxygen. Avoid using multiple layers of transparent drapes to secure dressings over intact skin, as this can decrease the transmission of moisture vapor and oxygen, which in turn may increase the risk of fungal infection, maceration, and loss of an intact seal.
Periwound maceration also may indicate increased wound exudate, requiring an increase in negative pressure. Conversely, an ecchymotic periwound may indicate excessively high negative pressures. If either occurs, assess the need to adjust negative pressure and intervene accordingly. Reassess NPWT effectiveness with subsequent dressing changes.
Apply a protective liquid skin barrier to the periwound and adjacent healthy tissue to help protect the skin surface from body fluids. The skin barrier also helps prevent stripping of fragile skin by minimizing shear forces from repetitive or forceful removal of transparent drapes. Excessive moisture can be absorbed by using a light dusting of ostomy powder sealed with a skin barrier. A “window pane” of transparent drape or hydrocolloid dressing around the wound also can protect surface tissue from contactwith NPWT material and prevent maceration.
Avoid creating rolled wound edges
In the best-case scenario, epithelial tissue at the wound edge is attached to the wound bed and migrates across healthy granulation tissue, causing the wound to contract and finally close. With deep wound environments that lack moisture or healthy granulation tissue, the wound edges may roll downward and epibole may develop. Epibole is premature closure of the wound edges, which prevents epithelialization and wound closure when it comes in contact with a deeper wound bed. (See Picturing epibole by clicking the PDF icon above.)
Materials used in NPWT are primarily air-filled. Applying negative pressure causes air removal, leading to wound contraction by pulling on the wound edges—an action called macrostrain. Without sufficient NPWT material in the wound, macrostrain can cause the wound to contract downward and the wound edges to roll.
Ensure that enough NPWT material has been applied into the wound to enhance wound-edge approximation and avoid creating a potential defect as the wound heals. Before NPWT begins, material should be raised 1 to 2 cm above the intact skin. Additional material may be needed with subsequent changes if the NPWT material compresses below the periwound. The amount of NPWT material needed to remain above the periwound once NPWT starts varies with the amount of material compressed and the wound depth.
Reduce the infection risk
To some degree, all wounds are contaminated. Usually, the body’s immunologic response is able to clear bacterial organisms and wound healing isn’t delayed. But a patient who has an infection of a complex wound needs additional support.
Systemic antibiotics alone aren’t enough because they’re selective for specific organisms and don’t reach therapeutic levels in the wound bed. In contrast, topical antimicrobial adjuncts, such as controlled-release ionic silver, provide broad-spectrum antimicrobial coverage against fungi, viruses, yeasts, and gram-negative and gram-positive bacteria, including methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci.
Consider using controlled-release ionic silver for a wound known to be infected or at risk for infection due to its location or potential urine or fecal contamination. To be bactericidal, ionic silver must be in concentrations of at least 20 parts per million; also, it must be kept moist and must come in direct contact with infected wound bed. At lower concentrations, organisms may develop resistance. Ionic silver has no known resistance or contraindications. Dressings using it come in several forms, including a hydrogel sheet, perforated sheet, cavity version, and semiliquid hydrogel. Be sure the form you choose doesn’t occlude the NPWT material and compromise therapy. (See NPWT for a patient with necrotizing fasciitis by clicking the PDF icon above.)
View: NPWT
Obtain a negative-pressure environment
One of the most daunting aspects of NPWT is obtaining and maintaining a good seal—in other words, avoiding the dreaded leak. Preventive skin measures may contribute to a poor seal; skin-care products containing glycerin, surfactant, or dimethicone may prevent adequate adhesion of NPWT drapes. Body oil, sweat, and hair may need to be minimized or removed.
To avoid leaks, don’t overlook the obvious—loose connections, a loose drainage collection canister, exposed NPWT material, and skinfolds extending beyond the transparent drape. Tincture of benzoin (with or without a thin hydrocolloid dressing) increases tackiness to enhance the adhesive property of a transparent drape on the diaphoretic patient and on hard-to-drape areas, such as the perineum. But be sure to use tincture of benzoin with discretion, as it may remove fragile periwound tissue when the dressing is removed.
Ostomy paste products can serve as effective filler. These pliable products can be spread into position to obtain a secure seal under the transparent drape in hard-to-seal areas, such as the perineum. Pastes remain flexible and can be removed without residue. Temporarily increasing NPWT pressure to a higher setting may help locate a subtle leak or provide enough negative pressure to self-seal the leak. Once the leak resolves, remember to return the pressure to the ordered setting.
Knowledge optimizes healing
It’s important to be aware of potential complications of NPWT (See Take care with NPWT by clicking on the PDF icon above). However, when applied correctly, NPWT is an effective option for managing complex wounds. Recognizing and managing potential complications at the wound site, ensuring periwound protection, minimizing epibole formation, and preventing wound infection can result in a better-prepared wound bed and promote optimal healing.
View: NPWT case study
Selected references
Baranoski S, Ayello EA. (2012). Wound Care Essentials: Practice Principles. 3rd ed. Springhouse, PA; Lippincott Williams & Wilkins.
Bovill E, Banwell PE, Teot L, et al. Topical negative pressure wound therapy: a review of its role and guidelines for its use in the management of acute wounds. Int Wound J. 2008;5:511-529.
Sussman C, Bates-Jensen B. Wound Care: A Collaborative Practice Manual for Health Professionals. 4th ed. Baltimore, MD; Lippincott Williams & Wilkins; 2011.
Ronald Rock is an Adult Health Clinical Nurse Specialist in the Digestive Disease Institute at the Cleveland Clinic in Cleveland, Ohio.
Read MoreClinical Notes
2012 guideline for diabetic foot infections released
Foot infections in patients with diabetes usually start in a wound, most often a neuropathic ulceration. So clinicians can better manage diabetic foot infections, the Infectious Diseases Society of America (IDSA) published “2012 Infectious Diseases Society of America Clinical Practice Guideline for the Diagnosis and Treatment of Diabetic Foot Infections” in the June 15 Clinical Infectious Diseases.
The guideline updates IDSA’s 2004 diabetic foot infections guideline. It focuses on appropriate therapy, including debridement of dead tissue, appropriate antibiotic therapy, removing pressure on the wound, and assessing (and potentially improving) blood flow to the foot. The guideline also provides suggestions regarding when and how long antibiotics should be administered for soft-tissue and bone infections.
When diagnosing a diabetic patient with foot infection, the guideline recommends clinicians evaluate the patient at three levels—the patient as a whole, the affected foot or limb, and the infected wound. The guideline also provides advice on when and how to culture diabetic foot wounds.
Access a podcast on the guideline, which is available in a smartphone format and as a pocket-size quick-reference edition.
Combining bariatric surgery with medical therapy improves glycemic control
In obese patients with uncontrolled type 2 diabetes, bariatric surgery and 12 months of medical therapy significantly improved glycemic control compared to those who received only medical therapy, according to a study in The New England Journal of Medicine. “Bariatric surgery versus intensive medical therapy in obese patients with diabetes” was a randomized, nonblinded, single-center trial that included 150 patients in three groups: medical therapy only, medical therapy and Roux-en-Y gastric bypass, and medical therapy and sleeve gastrectomy.
Although glycemic control improved for all three groups, those who received bariatric surgery had better control. Use of drugs to lower glucose, lipid, and blood-pressure levels decreased significantly after both surgical procedures but increased in patients receiving medical therapy only. No deaths or life-threatening complications occurred.
HHS launches web-based tool for tracking healthcare performance
The U.S. Department of Health and Human Services (HHS) has launched a web-based tool for monitoring the performance of the healthcare system. The Health System Measurement Project gives providers and the public the ability to examine datasets from across the federal government that span specific topic areas, such as access to care, vulnerable populations, prevention, and quality. Users can also view indicators by population characteristics, such as age, sex, income level, insurance coverage, and geography.
PEG tubes may increase risk of new pressure ulcers
According to a study published in Archives of Internal Medicine, percutaneous endoscopic gastrostomy (PEG) tubes may increase the risk of pressure ulcers in nursing home patients with advanced cognitive impairment.
Researchers found that hospitalized patients who receive a PEG tube were 2.27 times more likely to develop a new pressure ulcer and those with a pressure ulcer were less likely to have it heal when they had a PEG tube. “Our findings regarding the risk of developing new stage 2 or higher pressure ulcers suggest that PEG feeding tubes are not beneficial, but in fact they may potentially harm patients,” conclude the researchers in “Feeding tubes and the prevention or healing of pressure ulcers.”
AHRQ provides QI toolkit for hospitals
The Agency for Healthcare Research and Quality (AHRQ) offers a toolkit designed to help hospitals understand AHRQ’s quality indicators (QIs). “AHRQ Quality Indicators™ Toolkit for Hospitals” includes steps for improvement, how to sustain change, and different tools for different audiences. Clinicians can also access audio interviews that provide information on how to use the tools and engage stakeholders and staff in QI efforts, and a recording of a webinar on the toolkit.
Silk fibers may be future resource for bone and tissue repair
Researchers at Tufts University have developed the first all-polymeric bone scaffold material that is fully biodegradable and capable of providing significant mechanical support during repair. The material could improve the way bones and tissues are repaired after an accident or following disease effects.
The new technology uses micron-size silk fibers to reinforce a silk matrix, much as steel rebar reinforces concrete. The study, “High-strength silk protein scaffolds for bone repair,” published in Proceedings of the National Academy of Sciences, found that the scaffold material is significantly less strong than normal bone, but it may play a role as a temporary biodegradable support for the patient’s cells to grow.
International guidelines for silver dressings in wounds released
June’s Wounds International includes “International consensus: Appropriate use of silver dressings in wounds.”
A meeting of an international group of experts, convened by Wounds International, met in December 2011 to compile the consensus guidelines, which describe the patients who are most likely to benefit from silver dressings and how to use the dressings appropriately.
The guidelines recommend that silver dressings be used “in the context of accepted standard wound care for infected wounds or wounds that are at high risk of infection or reinfection.” Another recommendation is to use silver dressings for 2 weeks, then evaluate the wound, patient, and management approach before deciding whether to continue using the dressing or if a more aggressive intervention such as antibiotics would be better.
Cell therapy may benefit patients with lower extremity CLI
Injections of ixmyelocel-T in patients with lower extremity critical limb ischemia (CLI) who aren’t candidates for revascularization can prolong the time until treatment failure, according to a study in Molecular Therapy. Time to treatment failure was defined as major amputation, all-cause mortality, doubling of total wound surface area from baseline, or de novo gangrene. The double-blind, placebo-controlled RESTORE-CLI trial found that the adverse event rates were similar in the two groups.
New skin patch destroys skin cancer cells
A new skin patch destroyed facial basal cell carcinoma cells in 80% of patients, according to a study reported at the Society of Nuclear Medicine’s 2012 Annual Meeting.
Each of the 10 patients with facial basal cell carcinoma received a custom-made and fully sealed phosphorus-32 skin patch, a radiation spot-treatment in the form of a patch. Each patient was treated for 3 hours on the first day; the patches were reapplied on the fourth and seventh days after the first treatment for another 3 hours each. Three years after treatment, 8 of 10 patients were cancer-free.
The patients had lesions near the eyes, the nose, and forehead—areas more difficult to operate on, especially if skin grafting is needed later.
Small study links lymphedema to obesity
The average body-mass index (BMI) in obese patients with lymphedema was significantly greater than BMIs of obese patients without lymphedema, according to correspondence in The New England Journal of Medicine. The authors conclude, “Our findings suggest that obesity…may be a cause of lower-extremity lymphedema.”
“Lower-Extremity Lymphedema and Elevated Body-Mass Index” included 15 obese patients with bilateral lower-extremity enlargement who were referred to the authors’ center. Of the 15, five were diagnosed with lymphedema by lymphoscintigraphy.
Read MoreLearning to love your job
By Joan C. Borgatti, MEd, RN
The alarm clock goes off too early, and you jump-start the day with a cup of coffee and a short stack of reasons why you hate your job. Sound familiar? Although you can’t expect to love every aspect of your job, you should expect to get some degree of fulfillment from your career. If you don’t, maybe your job isn’t the problem. Maybe you just need a little career resuscitation to turn things around. First, let’s be clear. I’m not urging you to stay in a job that exposes you to unsafe conditions, a toxic environment, or a toxic boss. Call the code and get out, because emotional and physical well-being comes first. However, know that blaming our jobs for our dissatisfaction may be easier than taking a closer look at the chaos in our lives. It’s even easier not to fix what’s wrong, instead consoling ourselves with the company of like-suffering people. And misery does love company.
If you can’t have the job you love, love the job you have. The daily grind of Herculean demands can wear down even the most conscientious clinicians—to the point where we’re no longer seeking job satisfaction but struggling just to make it through the day. But you can turn things around. To enhance your job satisfaction, try these sure-fire methods. (Okay, maybe they’re not sure-fire, but they’re sure worth a try.)
Know when to say no
When your life feels out of balance, any demand will feel as if it’s sucking the living daylights out of you. You’ll be tempted to blame your job, when the truth is you’re giving in to a bottomless pit called “trying to please everyone else.” Learn to say no to the things you don’t want and say yes to more of what you do want. Say no to anything that’s not a priority (making cupcakes for the second-grade class). Say yes to quality time with your family and quality time for you (that painting class you’ve always wanted to take). Key question: How would the quality of your life improve if you started to say no to demands that don’t enhance its quality, and say yes to the things you want more of?
Learn to see the big picture again
Recognize that, in ways you can’t see or perhaps even imagine, you’ve forever touched and changed the lives of the patients you’ve cared for. The ability to touch and heal another person is a gift that’s available to few people in other professions, who struggle to find meaning in what they do. Key questions: In what ways have you helped your patients? What special qualities and skills are uniquely yours to give? How can you make the most of the opportunity to make a difference in patients’ lives?
Attract the positive
When we’re miserable, other miserable people gravitate to us. Soon a collective mindset takes root and the negative “group think” becomes a life-form unto
itself, festering and insatiable. So be careful of the company you keep. Surround yourself with positive people—clinicians committed to making a difference. This will reenergize you and give you a new perspective on your job.
Learn to be what you want
To be more passionate about your job,
focus on the aspects of the job that excite you the most. Passion is an energy form that attracts more of the same. Say, for instance, you’d love to buy a red convertible. One day you go out for a drive and you see red convertibles everywhere! Have more red convertibles suddenly driven off the assembly line? No; your mind is preselecting, or noticing the convertibles, for you. In the same way, you can preselect either more passion or more misery.
Pay it forward
Keep in mind that novice clinicians proceed through a learning curve. Rather than moan about how inexperienced they are, take one under your wing and turn her or him into the sort of clinician you’d want at your bedside if you were ill. You’ll rediscover your profession through this clinician’s eyes.
Communicate cleanly and ask for what you want
People can’t read your mind. To get more of what you want and less of what you don’t want, learn to communicate in a clean, neutral way. Let’s say you consistently wind up with the more difficult patient assignments. And let’s assume your boss does that because you’re the most clinically experienced clinician—not because she’s the devil incarnate. You can respond in one of two ways.
• Gripe to a coworker: “Can you believe she gave me that workload again?”
• Communicate with your boss cleanly and neutrally: “Lately it seems you’ve given me the more difficult patient assignments, and I appreciate your faith in me. Is there some way we can give other clinicians a chance to gain more experience caring for difficult patients? I’d be happy to act as a resource for them.”
See the difference? The first response does nothing to change the situation; it simply fuels the collective misery mindset. The second response communicates to the boss in a respectful, appreciative way (yes, bosses need appreciation, too!) and seeks a solution that pleases everyone.
Take action and follow your STAR
Using the mnemonic device “STAR” can guide you toward actions that increase your job satisfaction.
Success on your terms. We all define success differently. If you grew up in a family of college professors, chances are the healthcare field didn’t fit your family’s definition of success; your job dissatisfaction may stem from your inner turmoil over not meeting your family’s expectations. To key into these expectations, recall the “you should” and “you ought to” messages you heard as a child.
Key question: Take a moment to think about what success in your career would look and feel like. Then complete this sentence: “I know I will be successful when I have/I am _________.”
True north as your guide. A large part of how we judge ourselves, our worth, our success, and our happiness hinges on how other people see us. But true success, true happiness, and true job satisfaction are determined from within, by your inner compass. The captain of a ship must always know where true north is, because it never changes (much like our core values). He must know the difference between true north and compass north. Unlike true north, compass north is affected by the earth’s magnetic pull. In life, compass north is the magnetic pull of “you should do this” and “you ought to do that” messages. For instance, if you’re a skilled wound care clinician but have always been particularly passionate about lymphedema, you may dislike your job. That’s because you’ve ignored your true north (inner truth) and given in to compass north (fear of walking away from those current skills, and so forth). Don’t be afraid to follow your true north.
Key question: What steps can you take right now that will move you closer to your true north?
Assess and understand who you are. Most of us can articulate what our strengths are. But that’s not enough. To get more enjoyment from your job, you must stretch and exercise your strengths and look for ways to use them. If the opportunities aren’t there, create them.
Let’s say you’re the one everyone turns to for help when there’s a patient with a lower extremity ulcer. To leverage that strength, offer to hold an education program.
Key questions: List your strengths, and then ask yourself: How can I leverage these? If you’re too humble to recognize your strengths, give yourself 20 lashes (figuratively speaking); then ask a trusted colleague, “What do you see as my strengths?”
Risk it all (within reason). When we play it safe, our lives and careers can be pretty dull. We’re meant to push the envelope and stretch our capabilities. It puts the juice back in our lives and helps us grow and feel more alive. Nothing shakes out the cobwebs and brings excitement back to your career more than taking a risk. With every risk comes the threat of failure, but know that failure is just another form of data that helps you readjust and move forward. Don’t give failure more power than your successes.
Key questions: If you weren’t afraid, what risks would you consider taking to enhance your career? What’s holding you back?
Embrace change
An Eastern saying goes something like this: You can stand by a river, but you can never put your feet in the same place twice. The river is your life. It’s not stagnant; it’s ever changing. Nothing in life stays the same—not personal circumstances, relationships, or careers. You aren’t the clinician you were 10 years ago or even last year. So tweak your professional life to better reflect the clinician you are today. With a little attention, you could make your job the career of your dreams.
Selected references
Bird J. Do you need to love your job? Not necessarily. www.worklifebalance.com/love-your-job.html. Accessed May 21, 2012.
Borgatti J. Frazzled, Fried…Finished? A Guide to Help Nurses Find Balance. Borgatti Communications; 2004. www.joanborgatti.com and www.booklocker.com.
Colvin C. How to love the job you’ve got. www.womentodaymagazine.com/career/lovejob.html. Accessed May 21, 2012.
Johnson Montesol S. How to love the job you’ve got. http://developmentcrossroads.com/2011/01/how-to-love-the-job-you%E2%80%99ve-got/. Accessed May 21, 2012.
Joan C. Borgatti, MEd, RN, is the owner of Borgatti Communications in Wellesley Hills, Mass., which provides writing, editing, and coaching services. You may e-mail her at [email protected].
Read MoreAsk the treatment expert: Should treatment nurses be certified?
Discovery Promises Unique Medicine for Treatment of Chronic and Diabetic Wounds
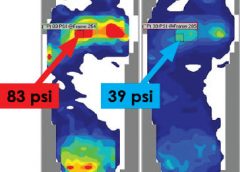
Pressure mapping: A new path to pressure-ulcer prevention
By: Darlene Hanson, MS, RN, Pat Thompson, MS, RN, Diane Langemo, PhD, RN, FAAN, Susan Hunter, MS, RN, and Julie Anderson, PhD, RN, CCRC
Faced with the nursing diagnosis of Impaired skin integrity, we’ve all written care plans that state our goal as “redistributing or reducing pressure.” But how do we do that? Which measures do we take? And how do we know that our interventions have relieved pressure? Do we rely solely on a skin assessment? A patient’s self-assessment of comfort? What if the patient can’t feel pressure relief because of neurologic impairment?
The answers to these questions may be that nurses should use pressure mapping, a tool used by occupational and physical therapists to determine seat-interface pressures and by other healthcare professionals to perform foot assessments. (more…)
Read More